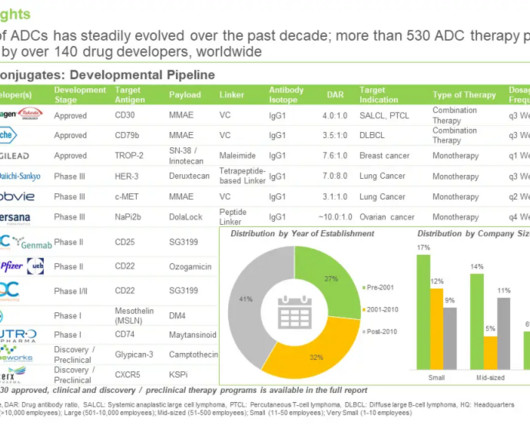
3BP and Novartis strike licensing agreement for FAP-targeting peptide technology
Pharmaceutical Technology
APRIL 25, 2023
German biotechnology firm 3B Pharmaceuticals (3BP) has entered into a licensing agreement with Novartis Innovative Therapies for its fibroblast activation protein (FAP)-targeting peptide technology. 3BP receives an initial payment of $40m, and $425m as development, regulatory and commercial milestone payments.




































Let's personalize your content