Mastering 21 CFR Part 11 Compliance: A Comprehensive Guide for Clinical Trials in the Digital Age
Cloudbyz
JUNE 10, 2023
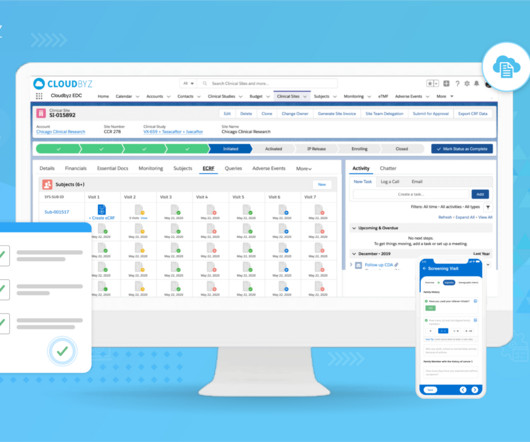
The Title 21 Code of Federal Regulations Part 11, or 21 CFR Part 11, is a regulation set forth by the FDA, which applies to all industries under its regulation, including pharmaceuticals, medical devices, biotechnology, and other life sciences. Ensure that the system properly authenticates users before granting access.









Let's personalize your content