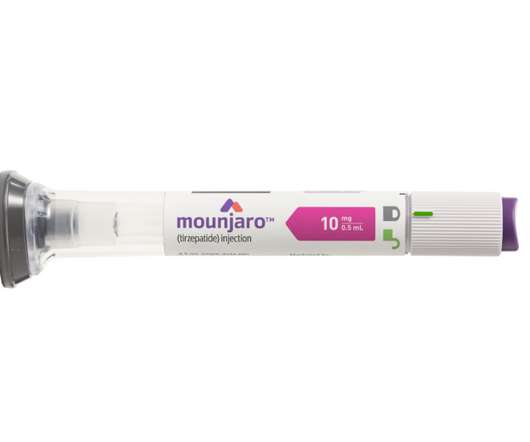
Lilly’s new drug Mounjaro (tirzepatide ) wins US FDA approval
pharmaphorum
MAY 16, 2022
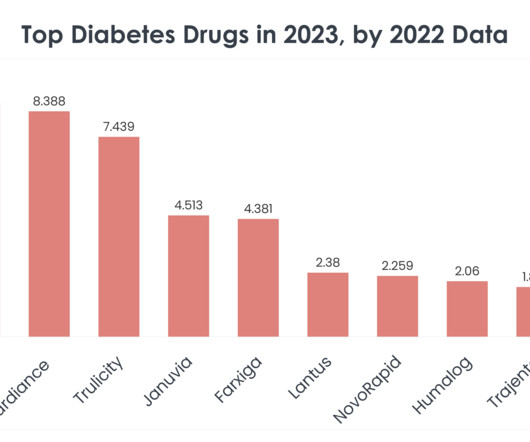
GLP-1 and GIP are hormones involved in blood sugar control and Mounjaro, a first-in-class medicine that activates both GLP-1 and GIP receptors, demonstrated improved blood sugar control. It was also compared to a placebo, a GLP-1 receptor agonist (semaglutide) and two long-acting insulin analogues. more than insulin degludec and 1.0%




















Let's personalize your content