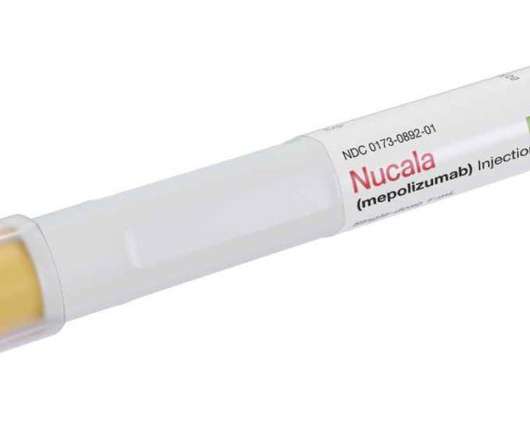
GSK announces FDA approval for Nucala (mepolizumab) for use in adults with chronic rhinosinusitis with nasal polyps
The Pharma Data
JULY 29, 2021
First IL-5 therapy approved as an add-on treatment in the US for adults with chronic rhinosinusitis with nasal polyps to target eosinophilic inflammation Fourth indication for mepolizumab in the US for eosinophil-driven diseases. It is not currently approved for use in COPD anywhere in the world. About mepolizumab. CONTRAINDICATIONS.























Let's personalize your content