GSK, Vir build case for COVID-19 antibody drug with new study data
Bio Pharma Dive
JUNE 21, 2021
Final clinical trial results confirmed treatment dramatically reduced the risk of hospitalization and death among adults with mild-to-moderate COVID-19.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

Bio Pharma Dive
JUNE 21, 2021
Final clinical trial results confirmed treatment dramatically reduced the risk of hospitalization and death among adults with mild-to-moderate COVID-19.

Pharmaceutical Technology
JULY 21, 2022
AstraZeneca has signed a deal with the Federal Office of Public Health (FOPH) of Switzerland to deliver over 1,200 doses of antibody therapy, tixagevimab and cilgavimab combination (AZD7442), for Covid-19 prevention and treatment. In June 2020, these antibodies, discovered at Vanderbilt University Medical Center, were licensed to AstraZeneca.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Bio Pharma Dive
NOVEMBER 14, 2024
The company is paying China-based biotech LaNova Medicines $588 million for the type of dual-pronged antibody drug that recently bested Keytruda in a clinical trial.
Bio Pharma Dive
JANUARY 20, 2021
Results from a clinical trial in nursing home residents and staff offered the first evidence Lilly's bamlanivimab could protect against, as well as treat, COVID-19

Pharmaceutical Technology
JANUARY 9, 2023
Eisai and Biogen have received approval for their antibody Leqembi (lecanemab-irmb) , 100mg/mL injection for intravenous use, from the US Food and Drug Administration (FDA) under the Accelerated Approval Pathway to treat Alzheimer’s disease (AD). The regulatory approval is based on the data obtained from the Phase II trial.

Pharmaceutical Technology
FEBRUARY 12, 2024
The humanised monoclonal antibody DISC-3405 is under investigation in a Phase I clinical trial, with data expected this year.

BioPharma Reporter
DECEMBER 13, 2021
Memo Therapeutics AG will receive CHF 10.5m (US $11.4m) from the Swiss government to clinically develop a SARS-CoV-2 antibody against COVID-19, allowing it to start Phase 1 studies in Q1, 2022.

pharmaphorum
JUNE 15, 2021
Antibody flunks post-exposure prevention study. Eli Lilly meanwhile reported positive results with its bamlanivimab antibody in the BLAZE-2 study carried put in nursing homes, which found a 57% reduction in the risk of infection compared to placebo.

Worldwide Clinical Trials
OCTOBER 14, 2024
Fortunately, advances in clinical research are providing hope for better treatments and outcomes. With the support of global networks like ours at Worldwide Clinical Trials (Worldwide), autoimmune and bone health research is accelerating, providing new solutions to patients around the world.

Pharmaceutical Technology
MAY 22, 2023
The Center for Drug Evaluation (CDE) of China’s National Medical Products Administration (NMPA) has accepted an investigational new drug application (IND) for SinoMab BioScience’s SM17 to treat asthma. SinoMab BioScience intends to commence a Phase I clinical trial in China to evaluate SM17’s safety profile.

Worldwide Clinical Trials
OCTOBER 14, 2024
Fortunately, advances in clinical research are providing hope for better treatments and outcomes. With the support of global networks like ours at Worldwide Clinical Trials (Worldwide), autoimmune and bone health research is accelerating, providing new solutions to patients around the world.

Pharmaceutical Technology
DECEMBER 27, 2022
Along the way, mergers and acquisitions continued to happen, new drugs and devices got approved, and innovations in the clinical trial industry were introduced. Drug pricing was a hot-button topic, and William Newton found that while e-commerce chains could cut insulin prices , any major change won’t happen overnight.

XTalks
JANUARY 21, 2025
The FDA has approved Datroway (datopotamab deruxtecan-dlnk), a TROP2-directed antibody-drug conjugate (ADC) for adults with unresectable or metastatic HR-positive, HER2-negative breast cancer, developed through a global collaboration between Daiichi Sankyo and AstraZeneca.
Pharmaceutical Technology
APRIL 14, 2023
Clinical-stage biopharmaceutical company TORL BioTherapeutics has raised $158m in a Series B financing round for advancing the development of new biologics for cancer treatment. TORL BioTherapeutics is a newly formed, US-based company focused on the development of novel antibody-based therapeutics for cancer patients. CLDN 18.2).
XTalks
MAY 2, 2022
This year, the monoclonal antibody lecanemab is approaching final stages of investigation as a treatment for targeting the A? The Phase IIb clinical trial focused on the long-term clinical outcomes and safety for patients with MCI and amyloid pathology (early AD). The main pathological markers of AD are beta-amyloid (A?)

Pharmaceutical Technology
FEBRUARY 6, 2023
It explores the monoclonal antibody detection that may work against procathepsin-L (pCTS-L), a pro-inflammatory protein mediator, as the potential remedy. These anti-pCTS-L monoclonal antibodies may help in providing effective treatments for human sepsis as well as other infectious diseases, such as Covid-19.

Pharmaceutical Technology
AUGUST 5, 2022
In addition, the lethality of prion diseases effectively means that any effective treatment could be fast-tracked into the clinic, streamlining the regulatory journey for companies, says Wille. No progress in drug discovery. “We There are only a few antibodies that can specifically recognize the (abnormal) version of the prion protein.

Pharmaceutical Technology
JULY 7, 2022
Brii Biosciences and TSB Therapeutics have commercially launched a long-acting neutralising antibody therapy combination, amubarvimab/romlusevimab, for Covid-19 in China. The two non-competing SARS-CoV-2 monoclonal neutralising antibodies, amubarvimab and romlusevimab are obtained from convalesced Covid-19 patients. 2 subvariants.

Pharmaceutical Technology
AUGUST 24, 2022
The global immunotherapy market, which includes CAR T-cell therapy, adoptive cell therapies, checkpoint inhibitors, monoclonal antibodies, and other immunotherapies, is expected to grow by 10.1% Patients provide blood samples for clinical trial sponsors to develop and deliver these emerging immunotherapies.

XTalks
DECEMBER 4, 2024
In this episode, Ayesha spoke with John Yu, MD, CEO of Kairos Pharma , a company advancing therapies to overcome challenges in cancer drug resistance and immune suppression. Kairos is developing therapeutics that reverse cancer drug resistance in cancers such as prostate and lung cancer. Dr. Yu has also led numerous clinical trials.

pharmaphorum
SEPTEMBER 17, 2020
Eli Lilly has followed up supportive data from its Olumiant in COVID-19 with results of a study showing its antibody treatment could help prevent hospitalisation from the disease. The double-blind placebo-controlled antibody study involved patients with symptoms outside hospitals, who were treated with four different dose strengths.

Medical Xpress
NOVEMBER 21, 2022
Over the past two decades, numerous complex cancer therapies called antibody drug conjugates (ADCs) have been tested in clinical trials and approved for use in patients. Their findings are published online in Cancer.

Pharmaceutical Technology
SEPTEMBER 16, 2022
The US Food and Drug Administration (FDA) has granted fast track designation to Jasper Therapeutics’ antibody, JSP191, for the treatment of severe combined immunodeficiency (SCID) patients who undergo allogeneic hematopoietic stem cell transplant. .

Bio Pharma Dive
AUGUST 20, 2024
In addition to new stock offerings, this week saw Merck license an antibody drug conjugate and BARDA further work trying to innovate remote clinical trials.
Pharmaceutical Technology
NOVEMBER 14, 2022
The US Food and Drug Administration (FDA) has granted accelerated approval for ImmunoGen’s Elahere (mirvetuximab soravtansine-gynx) to treat adults with folate receptor alpha (FR?)-positive, binding antibody, a cell-surface protein which is greatly expressed in ovarian cancer, as well as the maytansinoid payload DM4.

XTalks
NOVEMBER 12, 2024
Thus, to address drug-resistant infections, pharma and biotech companies are developing novel treatments such as bacteriophage therapies, immunomodulators and monoclonal antibodies. Monoclonal Antibody Therapies Monoclonal antibodies are gaining traction as targeted treatments for bacterial and viral infections.

Pharmaceutical Technology
AUGUST 8, 2022
AstraZeneca and Daiichi Sankyo ’s Enhertu (trastuzumab deruxtecan) has received expanded approval from the US Food and Drug Administration (FDA) to treat adults with unresectable or metastatic HER2-low (IHC 1+ or IHC 2+/ISH-) breast cancer. Enhertu-treated subjects had a median overall survival (OS) of 23.4 months compared with 16.8

Pharmaceutical Technology
AUGUST 12, 2022
The US Food and Drug Administration (FDA) has granted approval for Daiichi Sankyo and AstraZeneca ’s Enhertu (fam-trastuzumab deruxtecan-nxki) to treat unresectable or metastatic non-small cell lung cancer (NSCLC) in adults. In the trial, 1.9% According to the interim efficacy data from a pre-specified patient cohort, a 5.4

Pharmaceutical Technology
JUNE 2, 2023
Briumvi is an anti-CD20 monoclonal antibody indicated for RMS adult patients with active disease which is defined by clinical or imaging features. It has been engineered to remove certain sugar molecules generally expressed on the antibody. The therapy targets a unique epitope on CD20-expressing B-cells.

STAT News
DECEMBER 11, 2022
NEW ORLEANS — An antibody treatment developed by the Belgian drugmaker Argenx raised platelet counts and stopped bleeding episodes in patients with a rare autoimmune disorder that causes the body to attack and destroy its own blood-clotting platelets.

Bio Pharma Dive
MAY 9, 2022
Ameet Mallik takes over for founder Chris Martin at helm of the antibody drug developer after a seven-month run leading Rafael Holdings, which reported a clinical trial setback last October.
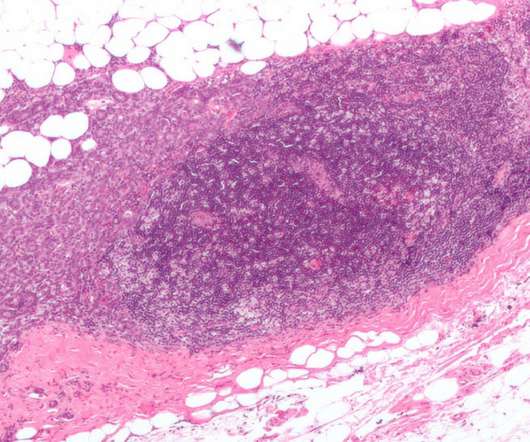
Pharmaceutical Technology
JANUARY 13, 2023
Being co-developed and co-marketed by Daiichi Sankyo along with AstraZeneca , Enhertu is a specifically engineered HER2-directed antibody-drug conjugate (ADC). Enhertu’s safety profile was consistent with the previous clinical trials.
Pharmaceutical Technology
NOVEMBER 18, 2022
The US Food and Drug Administration (FDA) has approved Provention Bio’s biologics licence application (BLA) for Tzield (teplizumab-mzwv) to treat type 1 diabetes (T1D) patients. The anti-CD3-directed antibody Tzield aims to delay the onset of Stage 3 T1D in adults and children aged eight years and above who are currently with stage 2 T1D.

pharmaphorum
FEBRUARY 8, 2024
Gilead Sciences has said it will no longer develop its CD47-targeting antibody magrolimab in blood cancers after data suggested that patients taking the drug in clinical trials were more likely to die than those in control groups.

Pharmaceutical Technology
MAY 8, 2023
Ei s ai and Bliss Biopharmaceutical have entered into a joint development agreement for antibody-drug conjugate (ADC), BB-1701, to treat cancers. Currently, BB-1701 is being evaluated in Phase I/II trials in China and the US. Currently, BB-1701 is being evaluated in Phase I/II trials in China and the US.

XTalks
APRIL 22, 2022
Flow cytometric receptor occupancy assays are being increasingly used in preclinical and clinical studies. Both the areas of drug development and clinical trials are increasingly using in vitro assays to help determine the efficacy of an investigational therapeutic. What is Flow Cytometry?

STAT News
JANUARY 10, 2023
SAN FRANCISCO — For months, drugmakers have been pleading with regulators to lower the bar for authorizing antibody drugs for Covid. The virus, they noted, had evolved fast enough to render every previous antibody obsolete.

XTalks
MAY 4, 2023
A blood test can detect antibodies to gluten, but a definitive diagnosis requires a biopsy of the small intestine. As of now, the US Food and Drug Administration (FDA) has not approved any drugs for the treatment of celiac disease. How does Beyond Celiac spread awareness about research and clinical trials for celiac disease?

STAT News
NOVEMBER 13, 2022
Roche reported negative results Monday from a pair of clinical trials investigating an antibody treatment for people with early-stage Alzheimer’s disease — another setback for the Swiss pharma giant’s decades-long effort to develop medicines against the memory-destroying disease.

Worldwide Clinical Trials
JUNE 21, 2024
The 2024 ASCO Annual Meeting from the American Society of Clinical Oncology is a fantastic platform for clinical researchers to discuss the latest advancements and challenges in oncology research. Currently, there are 12 approved ADC drugs, and several hundred more are in the development pipeline.
STAT News
NOVEMBER 28, 2022
… A 65-year-old woman who was receiving a promising experimental treatment to slow the cognitive decline caused by her early Alzheimer’s disease recently died from a massive brain hemorrhage that some researchers link to the drug , Science reports. STAT reported on another such case last month.

Pharmaceutical Technology
JANUARY 26, 2023
Health Canada has accepted for review Valeo Pharma partner Veru’s new drug submission (NDS-CV) for Covid-19 therapy sabizabulin. Valeo Pharma CEO Steve Saviuk said: “Health Canada’s filing acceptance of Veru’s NDS-CV application for sabizabulin is the first important regulatory milestone in the drug approval process.

Medical Xpress
APRIL 10, 2023
Repeat radiation therapy combined with the monoclonal antibody drug bevacizumab extended progression-free survival in patients with recurrent glioblastoma, according to findings from a recent clinical trial published in the Journal of Clinical Oncology.

Pharmaceutical Technology
JANUARY 8, 2023
In 2023, the pharmaceutical industry will mark 20 years since Xolair, an anti-IgE antibody, became the first biologic approved to treat asthma. Since then, the US FDA, EMA, and other agencies have approved several biologic antibodies targeting the inflammatory cytokines IL-4, IL-13, IL-5, and others for asthma. Ongoing efforts.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content