Avidity, Kymera raise fresh funds; Walgreens partners with BARDA
Bio Pharma Dive
AUGUST 20, 2024
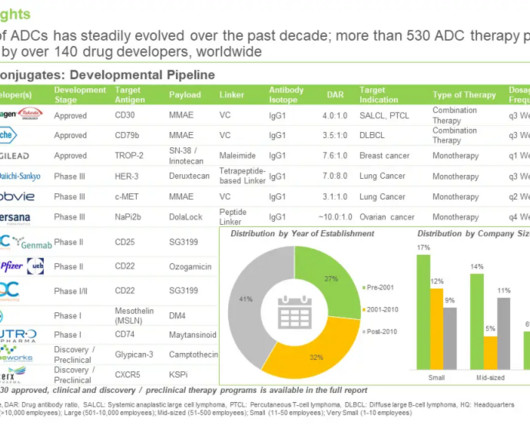
In addition to new stock offerings, this week saw Merck license an antibody drug conjugate and BARDA further work trying to innovate remote clinical trials.



































Let's personalize your content