US FDA grants Fast Track status for Avidity’s AOC 1020 to treat FSHD
Pharmaceutical Technology
JANUARY 19, 2023
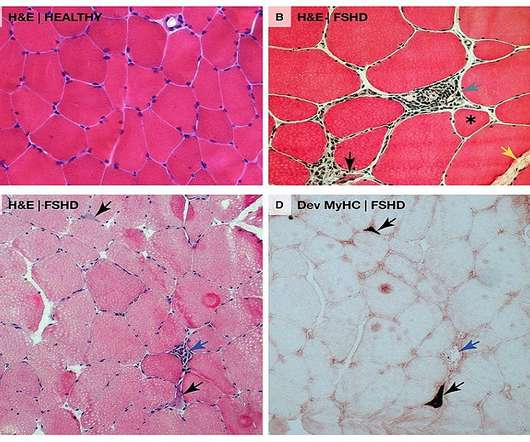
This DUX4 protein abnormal expression leads to modifications in gene expression in muscle cells which are associated with progressive muscle function loss in FSHD patients. Avidity Biosciences’ AOC 1020 is intended to reduce the DUX4 mRNA and DUX4 protein expression in muscles in these patients.


































Let's personalize your content