Health Canada gives approval to Enhertu for breast cancer treatment
Pharmaceutical Technology
JANUARY 13, 2023
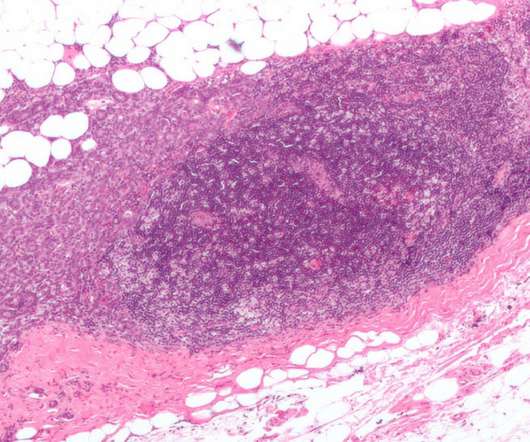
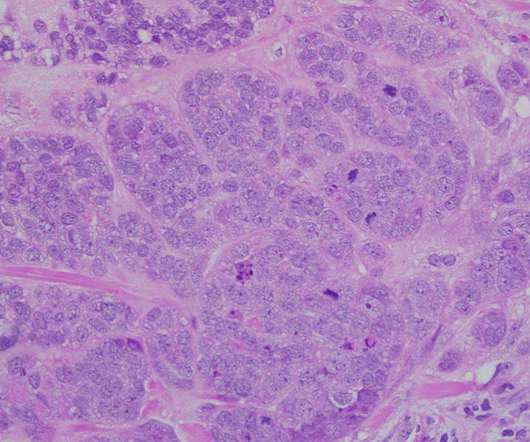
Being co-developed and co-marketed by Daiichi Sankyo along with AstraZeneca , Enhertu is a specifically engineered HER2-directed antibody-drug conjugate (ADC). Enhertu’s safety profile was consistent with the previous clinical trials.





































Let's personalize your content