ASH23: Pharma branding, Editas’ high bar and clinical trial diversity
Bio Pharma Dive
DECEMBER 11, 2023
Editas had the tall task Monday of convincing ASH attendees its gene therapy for sickle cell disease could improve on Casgevy and Lyfgenia.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

Bio Pharma Dive
DECEMBER 11, 2023
Editas had the tall task Monday of convincing ASH attendees its gene therapy for sickle cell disease could improve on Casgevy and Lyfgenia.

AuroBlog - Aurous Healthcare Clinical Trials blog
NOVEMBER 14, 2024
The Subject Expert Committee (SEC), which advises the national drug regulator on matters related to approval of new drugs and medical devices and clinical trials, has recommended grant of market authorisation for Telangana-based MSN Laboratories for sleep disorder drug Pitolisant tablets.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Drug Patent Watch
DECEMBER 12, 2024
Identifying branded drugs with a low likelihood of generic entry has become a crucial strategy for companies looking to expand their product portfolio through in-licensing. Branded drugs, developed through extensive research and clinical trials, often enjoy patent protection for a limited period.

AuroBlog - Aurous Healthcare Clinical Trials blog
NOVEMBER 22, 2022
Drug manufacturers have hailed the Drugs (Eighth Amendment) Rules, 2022 mandating barcode or quick response (QR) code on the label of top 300 brands of formulations from August 1, 2023, saying that QR codes will help identify misbranded or counterfeit medicines as well as recall the products if its quality gets compromised during manufacturing.
Deltaclinical
NOVEMBER 2, 2021
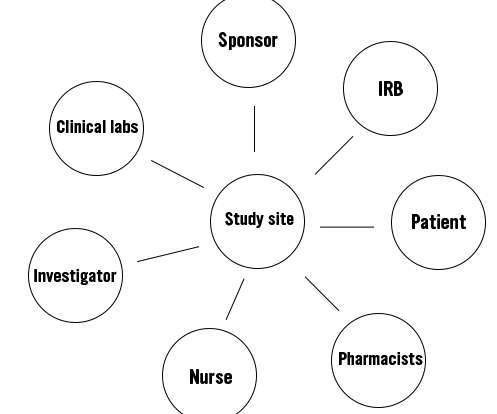
Along with a team of nurses and other stakeholders, the PI will conduct the trial at his/her study site. However, the sponsor will remain the monitor of the trial. The next figure shows the important stakeholders in a clinical trial. Traditionally, clinical trials have never been designed with patient-centricity in mind.

Deltaclinical
NOVEMBER 2, 2021
Along with a team of nurses and other stakeholders, the PI will conduct the trial at his/her study site. However, the sponsor will remain the monitor of the trial. The next figure shows the important stakeholders in a clinical trial. Traditionally, clinical trials have never been designed with patient-centricity in mind.

Deltaclinical
NOVEMBER 2, 2021
Along with a team of nurses and other stakeholders, the PI will conduct the trial at his/her study site. However, the sponsor will remain the monitor of the trial. The next figure shows the important stakeholders in a clinical trial. Traditionally, clinical trials have never been designed with patient-centricity in mind.

Deltaclinical
NOVEMBER 2, 2021
Along with a team of nurses and other stakeholders, the PI will conduct the trial at his/her study site. However, the sponsor will remain the monitor of the trial. The next figure shows the important stakeholders in a clinical trial. Traditionally, clinical trials have never been designed with patient-centricity in mind.

AuroBlog - Aurous Healthcare Clinical Trials blog
SEPTEMBER 16, 2024
Sold under the brand name Ubrelvy, ubrogepant is already used to treat migraines, but researchers in the US wanted to […]

World of DTC Marketing
OCTOBER 21, 2021
SUMMARY: Medicare spent nearly $600 million over a three-year period to pay for four cancer drugs with no clinical benefit an analysis published Monday by JAMA Internal Medicine found. billion, with more than half of that devoted to products that did not provide a “documented overall survival benefit” in clinical trials.

AuroBlog - Aurous Healthcare Clinical Trials blog
OCTOBER 15, 2024
(Abdullah Durmaz/Getty Images) In just a few years, brand-name injectable drugs such as Ozempic, Wegovy, Mounjaro and Zepbound have rocketed to fame as billion-dollar annual sellers for weight loss as well as to control blood sugar levels and reduce the risk of heart disease.
XTalks
JANUARY 29, 2021
This is relevant for clinical trials as more regulators require evidence of efficacy in comparison to the standard of care, which is likely to be one of the blockbuster products. As more trials show equivalency in outcomes between the branded and biosimilar product, more purchasers will choose the cheaper option.

Clinical Trial Podcast
JULY 18, 2020
Have you been tasked to develop a clinical trial budget? Well, you’re in luck because I’m going to share everything you need to know about clinical trial costs. Clinical trial budgets are often put together in haste. Developing a clinical trial budget can be a confusing exercise for sponsors and CROs.
Cloudbyz
MARCH 17, 2021
Monitoring patient safety during a clinical trial is one of the founding principles to be followed throughout the drug development life cycle. It can be defined as a collaborative relationship between sponsors, sites, researchers, and everyone involved in the clinical trial phases.

AuroBlog - Aurous Healthcare Clinical Trials blog
NOVEMBER 17, 2022
Acetaminophen, also known as paracetamol and sold widely under the brand names Tylenol and Panadol, also increases risk-taking, according to a study from 2020 […]. One of the most consumed drugs in the US – and the most commonly taken analgesic worldwide – could do a lot more than simply take the edge off your headache.

pharmaphorum
NOVEMBER 13, 2020
Where a marketing company might own and manage a domain on behalf of a pharma brand, written proof from the brand is needed to confirm collaboration with the marketing company. The post Pharma brands must ‘up the ante’ in their digital presence appeared first on. Case Study: UK leading emergency contraceptive pill.
AuroBlog - Aurous Healthcare Clinical Trials blog
JULY 17, 2023
The pill, called Opill – the brand name for the tablet formulation of norgestrel – is an oral contraceptive containing only progestin hormone, which helps prevent pregnancy […]

pharmaphorum
FEBRUARY 11, 2021
Pharma companies face many challenges when involving patients in the design of clinical trials – but doing so can have huge benefits further down the line, improving the sustainability and quality of research. The post Increasing patient engagement with UK clinical trials appeared first on.

Pharmaceutical Technology
NOVEMBER 29, 2022
According to data from the ARASENS clinical trial carried out at nearly 300 sites globally, including various NHS hospitals, subjects who received the combination therapy had a 32.5% It is branded as Nubeqa and is already offered on the NHS for patients with localised prostate cancer.

AuroBlog - Aurous Healthcare Clinical Trials blog
NOVEMBER 2, 2022
Care-Keralam, the Ayush cluster in Kerala, will shortly fine-tune its strategies to build ‘Kerala Brand of Ayurveda Products’ for international marketing and to increase the quantity of exports of traditional and herbal drugs from Kerala to global markets.

WCG Clinical
JUNE 12, 2023
June 12, 2023 — WCG, one of the world’s leading providers of solutions that measurably improve the quality, efficiency, and safety of clinical research, today announces the launch of its new brand identity. In 2022, we touched 90 percent of all clinical trials globally. That’s the beauty of our new brand.”

AuroBlog - Aurous Healthcare Clinical Trials blog
MARCH 17, 2024
The Department of Pharmaceuticals (DoP) has issued a new Uniform Code for Pharmaceutical Marketing Practices (UCPMP) 2024, permitting pharma companies to provide brand reminders such as informational and education items and free samples to medical professionals with restrictions on sample packs and total value.

AuroBlog - Aurous Healthcare Clinical Trials blog
JUNE 25, 2023
The Department of Pharmaceuticals (DoP) has rejected review applications filed by the Maharashtra-based Bharat Serums and Vaccines Ltd against National Pharmaceutical Pricing Authority (NPPA) fixing the ceiling price of its infertility treatment drug brand Hucog HP, since the company did not file the petition within the prescribed time limit.

XTalks
MARCH 12, 2025
This edition delved into the challenges and opportunities related to diversity in clinical trials, an essential aspect of modern clinical research that aims to ensure equitable representation and improved outcomes for all populations. DHTs : How digital tools are improving trial efficiency and accessibility.

pharmaphorum
MAY 20, 2022
Last year, the company added clinical trials to the list with the launch of CVS Health Clinical Trial Services , an initiative that works with pharma companies on clinical trial recruitment and even hosts trial sites at certain retail locations. This interview has been edited for length and clarity.

BioPharma Reporter
SEPTEMBER 3, 2020
Bluebird bio has reported end points were met in a clinical trial involving its candidate ALD cell therapy, branded as Lenti-D.

XTalks
MARCH 26, 2025
This enhances brand recognition while also attracting new clients, strategic partners and speaking opportunities that further solidify their leadership. Our Thought Leadership Checklist outlines the essential steps to position your brand as an authority in clinical research.

Drug Patent Watch
DECEMBER 30, 2024
Types of Drug Applications The PMDA accepts three main types of drug applications: Investigational New Drug (IND) : Required for conducting clinical trials in Japan. The ANDA must demonstrate bioequivalence to the Reference Listed Drug (RLD), which is the original brand-name drug approved for marketing in Japan.

World of DTC Marketing
OCTOBER 11, 2021
1ne: Don’t believe that TV alone will drive brand objectives; it’s not. They’re brighter and want to understand the data behind clinical trials better. If you plan to launch a new drug with the model of heavy-up TV and a website with some programmatic ads, you’re going to fail. 7even : Beware the PR trap.

XTalks
MARCH 19, 2025
Over the years, Xtalks has built the industrys preeminent life science podcast, helping brands in pharma, biotech, healthcare and medical devices showcase their expertise to key decision-makers through valuable, industry-leading content. Dont miss out on this opportunity to elevate your brand and stay top-of-mind in a competitive market.

Pharma Mirror
SEPTEMBER 11, 2022
Bandung, W Java, Indonesia, PT Bio Farma, the holding company for state-owned pharmaceutical companies in Indonesia, announced a new milestone in the manufacturing of IndoVac, a Covid-19 vaccine brand it has developed since November 2021.

Pharma Mirror
OCTOBER 1, 2022
Clinical Edge trains and certifies visual function examiners at investigator sites conducting Phase I to IV ophthalmic clinical trials. The combination of Optym, Emmes’ ophthalmology certification unit, and Clinical Edge will make the organization a leader in ophthalmic certification and training services.

XTalks
OCTOBER 24, 2024
Medidata , a Dassault Systèmes brand and leading provider of clinical trial solutions to the life sciences industry, announced Medidata Rave Lite , an extension of the company’s gold-standard clinical research software, Medidata Rave EDC , designed explicitly for Phase I and Phase IV studies.

World of DTC Marketing
FEBRUARY 7, 2022
If they decide to continue with clinical trials, a neutral, independent third party needs to review the data. Biogen used to be known as an innovative Biotech company; now, their brand is associated with misinformation and desperation.
Pharmaceutical Technology
MAY 1, 2023
The NDA is supported by the data package licensed to Ocumension by Nicox as well as the Phase III clinical trial in China. Zerviate, 0.24% was compared to emedastine difumarate ophthalmic solution, 0.05%, an antihistamine, which is marketed under undeb Emadine brand name.
Pharmaceutical Technology
SEPTEMBER 14, 2022
It is marketed under the brand name Covovax in the country. The latest development was based on the totality of manufacturing, preclinical and clinical trial findings filed for assessment. This data also comprises two Phase III clinical trials: a trial in the UK and PREVENT-19 in the US and Mexico.

Pharma Marketing Network
OCTOBER 7, 2024
In the fiercely competitive pharmaceutical industry, effective brand positioning is crucial for marketers striving to distinguish their products and services. With an ever-evolving market landscape, pharmaceutical companies must develop robust strategies to position their brands distinctively.

Pharmaceutical Technology
FEBRUARY 24, 2023
The overhaul will address drug marketing exclusivity length, pricing, patient access, innovation incentives, antimicrobial resistance, clinical trials, supply chain security and shortages, and environmental impact. The EU is planning a sweeping revision of its pharma legislation in March, the largest change in 20 years.

Roots Analysis
MARCH 14, 2023
Clinical trials are a fundamental requirement for evaluating the safety and efficacy of novel therapeutic interventions. Initiating a clinical trial is a complex and time-consuming process, involving suitable planning and execution.

Pharma Marketing Network
FEBRUARY 27, 2025
Introduction In todays digital-first world, content marketing campaigns are no longer optional for pharmaceutical brands. Imagine content marketing as the heartbeat of a pharma brands digital presence. Instead of traditional promotional tactics, pharma brands can use educational content to build credibility.

Drug Patent Watch
MARCH 26, 2025
Generic drugs have long been a vital part of this equation, offering a more accessible alternative to brand-name medications. From navigating complex regulatory landscapes to ensuring the integrity of clinical trials, the stakes are high for generic drug manufacturers.

pharmaphorum
JANUARY 12, 2022
Biogen’s difficult launch of controversial Alzheimer’s therapy Aduhelm has been made even harder by a proposal by Medicare to cover the drug only for certain patients enrolled in clinical trials. The post Medicare limits Aduhelm coverage to clinical trial participants appeared first on.

World of DTC Marketing
APRIL 27, 2021
Some Oncologists I spoke to said they want to hear what’s been going on with new drug development and ongoing clinical trials, but for PCPs, it might be “take a number and wait.” The problem is that physicians are overwhelmed with returning patients and often don’t have the time to meet with reps.

World of DTC Marketing
MAY 16, 2022
The drug led to impressive blood sugar and body weight drops in clinical trials. The approval for tirzepatide—which Lilly will market under the brand name Mounjaro—isn’t for weight loss but Type 2 diabetes. Food and Drug Administration approved Eli Lilly’s novel treatment for diabetes.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content