Cardiovascular Clinical Trials with Patient Diversity: Challenges and Considerations
XTalks
AUGUST 4, 2022
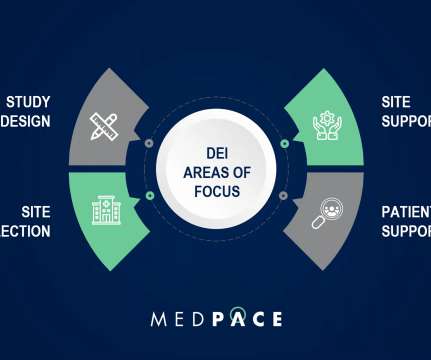
This started back in 1993 when the FDA established guidelines to increase diversity by gender and race/ethnicity of participants in clinical trials that were contributing to new drug approvals. Lack of diversity is more pronounced in industry-funded trials. or greater than 1.2








Let's personalize your content