AstraZeneca automates DNA synthesis to slash costs and timelines
BioPharma Reporter
FEBRUARY 6, 2023
AstraZeneca has cut the cost and time needed to complete DNA synthesis by integrating an assembly framework into Benchling.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

BioPharma Reporter
FEBRUARY 6, 2023
AstraZeneca has cut the cost and time needed to complete DNA synthesis by integrating an assembly framework into Benchling.
Outsourcing Pharma
JULY 18, 2023
company KSQ Therapeutics to co-develop KSQâs lead cancer drug, which is designed to sabotage the DNA repair mechanisms of cancer cells. Roche has entered a licensing and collaboration deal with the U.S.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Camargo
JULY 27, 2021
When developing a clinical development program for these increasingly popular therapies, it is important that sponsors use modeling and pharmacokinetic (PK) analysis to evaluate parameters that can be measured while dosing with gene therapy drugs, to characterize exposure-response data and inform rational dosing.

Scienmag
APRIL 22, 2021
Dose-sparing regimens and intradermal delivery have important implication for rapid clinical development of effective, well-tolerated and easy-to-distribute vaccines against MERS and other emerging coronaviruses.

Pharmaceutical Technology
MAY 17, 2023
Precision oncology company Boundless Bio has raised $100m in a Series C financing round for advancing extrachromosomal DNA (ecDNA)-directed therapies for oncogene-amplified cancer patients.
Pharmaceutical Technology
MAY 10, 2023
By adding an additional pipeline targeting oncology and inflammation diseases, the company expects that the deal will complement its existing clinical development priorities.

Pharmaceutical Technology
AUGUST 5, 2022
A PARP inhibitor, Lynparza (olaparib) is said to be the first targeted therapy to hinder DNA damage response in cells/tumours with a deficiency in homologous recombination repair. The US Food and Drug Administration (FDA) approved Lynparza in March for this indication.

Pharmaceutical Technology
MARCH 1, 2023
Nedisertib is under clinical development by Merck and currently in Phase I for Neuroendocrine Tumors. The drug candidate targets DNA-dependent protein kinase (DNA-PK). It was also under development for small cell lung cancer, rectal cancer and chronic lymphocytic leukemia (CLL). It is a new chemical entity.

Pharmaceutical Technology
MARCH 1, 2023
Nedisertib is under clinical development by Merck and currently in Phase I for Head And Neck Cancer Squamous Cell Carcinoma. The drug candidate targets DNA-dependent protein kinase (DNA-PK). It was also under development for small cell lung cancer, rectal cancer and chronic lymphocytic leukemia (CLL).

Worldwide Clinical Trials
JULY 8, 2024
With radiopharmaceuticals emerging as a diagnostic and therapeutic (theranostics) procedure, many are in the clinical development pipeline and are expected to play a crucial role in the future of healthcare. Genetic Factors : Certain genetic mutations, such as those in DNA repair genes, can affect how cells respond to radiation.

The Pharma Data
OCTOBER 21, 2020
and Shionogi Limited as shareholders, today announced the positive findings of a pooled analysis of COVID-19-related impacts across the investigational long-acting cabotegravir and rilpivirine clinical development programme.

XTalks
NOVEMBER 6, 2020
The Burnaby, BC-based company developed the oral DNA-based vaccine using its proprietary bacTRL Gene Therapy Platform, which uses genetically modified bifidobacteria as carriers of genetic vaccine elements on a DNA plasmid. Related: Red Meat Allergy Test Gets FDA Clearance. “We

pharmaphorum
APRIL 22, 2022
The write-down comes in the wake of problems affecting the Japanese drugmaker’s AT132 gene therapy candidate for rare disease X-linked myotubular myopathy (XLMTM), which was placed on clinical hold by the FDA last year after four patient deaths liked to possible liver-related side effects.

Pharmaceutical Technology
MARCH 2, 2023
Rituximab biosimilar is under clinical development by Dr. Reddy’s Laboratories and currently in Phase III for Rheumatoid Arthritis. It is produced using recombinat DNA technology in Chinese Hamster Ovary (CHO) cells. Reditux is indicated for the treatment of non-Hodgkin lymphoma.
pharmaphorum
FEBRUARY 22, 2021
Sanofi and GlaxoSmithKline are restarting clinical development of their COVID-19 vaccine, which was delayed after hitting a snag late last year. Sanofi’s shot combines the recombinant DNA technology it uses to make flue vaccines with the adjuvants GSK uses to boost the immune system with its jabs.

Drug Discovery Today
JULY 28, 2020
Cancer Research UK, the University of Southampton and Touchlight Genetics, a London based biotechnology company, today (Wednesday) announce a new clinical development partnership to progress a therapeutic DNA vaccine, TGL-100, into an early phase clinical trial targeting head and neck squamous cell carcinoma (HNSCC).

Pharmaceutical Technology
FEBRUARY 24, 2023
Amivantamab is under clinical development by Johnson & Johnson and currently in Phase I for Colorectal Cancer. Attributes of the drug, company and its clinical trials play a fundamental role in drug-specific PTSR and likelihood of approval. It is formulated as solution and concentrate for solution for intravenous infusion.
Roots Analysis
FEBRUARY 27, 2024
Over the last few decades, various viral vector manufacturing have been developed, optimized and standardized for introduction of therapeutic DNA / gene of interest into a patient’s body / cells. On the other hand, among non-viral gene delivery tools, plasmid DNA has emerged as the preferred option.

pharmaphorum
JANUARY 21, 2022
Pre-clinical studies demonstrate that inhibition of CK2 by Silmitasertib prevents DNA repair, induces apoptosis, and improves the antitumor activity of gemcitabine and cisplatin,” states Senhwa. There is a significant need for new BTC treatment options.”.

pharmaphorum
OCTOBER 29, 2020
BMS will have the option to select targets identified by insitro and then lead clinical development. The private, San-Francisco based company focuses on DNA sequencing technology. It synthesises, breeds and analyses large combinatorial chemicals that are encoded by DNA sequences called DNA-encoded libraries, or DEL’s.

Worldwide Clinical Trials
JUNE 15, 2022
Further, some patients are still hesitant to go into the clinic, and the prevalence of misinformation from the pandemic has led to greater concerns about therapies like gene therapies and the impact they fear it will have on their RNA/DNA. The cell and gene therapy pipeline is booming.

Delveinsight
DECEMBER 1, 2020
BASE10 Collaborates with DNA Link for COVID-19 Antibody Test Research. US-based BASE10 Genetics and Korean firm DNA Link announced a collaboration on a research project to appraise the usability of the DNA Link’s antibody test, AccuFind COVID-19 IgG, in a healthcare setting. CABOMETYX is already approved in the U.S.

The Pharma Data
MARCH 15, 2022
“There remains a significant unmet need for patients diagnosed with advanced prostate cancer, who have a poor prognosis after not responding to initial therapy,” said Dr. Roy Baynes, senior vice president and head of global clinical development, chief medical officer, Merck Research Laboratories.

pharmaphorum
JULY 5, 2021
The new deal will assists the company’s focus “on advancing and innovating Parexel to meet our customers’ needs across the evolving clinical development landscape,” he added. billion.
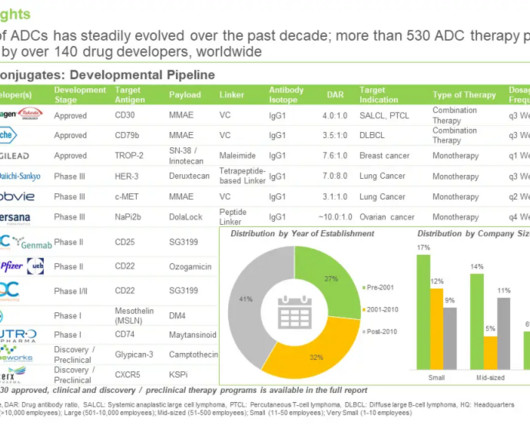
Roots Analysis
FEBRUARY 27, 2024
The firm also has the right to obtain exclusive licenses to the Affimer proteins for clinical development and commercialization. How many of the ADCs in development have pyrrolobenzodiazepine (PBD) as warhead: Pyrrolobenzodiazepines are a class of compounds that kill cells by binding their DNA and interfering with replication.
XTalks
JANUARY 19, 2024
When alpha-emitting radionuclides are delivered to cancer cells, the emitted alpha particles cause substantial damage to the DNA of the cells, leading to cell death. For example, non-target effects such as the release of damaged DNA into the cytoplasm of irradiated cells can lead to the activation of immune cells.
pharmaphorum
SEPTEMBER 8, 2022
However, there is a difference between the two: the toxic payloads of ADCs need to be absorbed into cancer cells, whereas radiopharmaceuticals can still do damage even when only bound to the cancer, and are particularly sensitive to radiation-induced DNA damage. Investment is flowing.

The Pharma Data
MAY 15, 2023
The acquisition complements Gilead’s existing clinical development priorities by adding additional pipeline assets for well-validated targets in oncology and inflammation. Both programs have the potential to address multiple indications, offering broad development opportunities alone and in combination with Gilead’s portfolio.

The Pharma Data
JANUARY 20, 2021
Nasdaq: INO), a biotechnology company focused on bringing to market precisely designed DNA medicines to treat and protect people from infectious diseases, including COVID-19, cancer and HPV-associated diseases, today announced the pricing of an underwritten public offering of 17,700,000 shares of its common stock at a public offering price of $8.50

The Pharma Data
DECEMBER 2, 2020
Results show the protocol demonstrated a 30Gb+ yield of long DNA reads raw data of an E. The FMv8 protocol introduces a needle-aspirate-based, cell-resuspension step prior to cell lysis that boosts DNA recovery. Elsewhere around the globe: RevoluGen – U.K.-based to establish a new joint venture company, Nanjing AuroRNA Biotech Co.,

The Pharma Data
OCTOBER 19, 2020
Caris Precision Oncology Alliance members also have access to the Caris Pharmatech oncology trial network, which can help reduce the time it takes to identify and connect appropriate patients with novel targeted cancer therapies in clinical development.

The Pharma Data
FEBRUARY 22, 2022
He is co-founder and non-executive board member of the Hartwig Medical Foundation (large scale DNA analyses) and is a board member of the Center for Personalized Cancer Treatment and leads several innovative precision oncology clinical trials. He began his oncology development career at Rhône Poulenc Rorer Inc.

The Pharma Data
AUGUST 6, 2020
In addition, Inovio previously teamed up with Beijing Advaccine Biotechnology for work on its DNA vaccine candidate, INO-4800. China has several home-grown COVID-19 vaccines in clinical development. Outside of China, BioNTech and its partner Pfizer launched their 30,000-subject phase 3 efficacy trial last week.

The Pharma Data
JANUARY 27, 2021
We are pleased to be advancing our second therapy into clinical development in our quest to bring transformative medicines to patients who need them,” said Bruce Goldsmith, Ph.D., We are excited to investigate the potential of PBFT02 as a treatment for FTD-GRN as we initiate our clinical development program in the coming months.”.

pharmaphorum
MARCH 8, 2022
DI: Oncology is a hugely exciting area at the moment, particularly the further focus on genome testing to help identify specific mutations in the DNA and select the best treatment option for the patient and their type of cancer. Avideh Nazeri is vice president clinical development, medical, and regulatory affairs, Novo Nordisk UK.

The Pharma Data
JANUARY 21, 2021
Session: Tumor Biology: Focus on EGFR Mutation, DNA Repair and Tumor Microenvironment Mini Oral Session Date and Time: January 31, 2021, 17:20 SGT (January 31, 2021 1:20 a.m. Spectrum Pharmaceuticals is a biopharmaceutical company focused on acquiring, developing, and commercializing novel and targeted oncology therapies.

The Pharma Data
JUNE 7, 2023
Acuitas’ proprietary LNP technology is used in multiple vaccines and therapeutics in clinical development and was also used in some of the COVID-19 vaccines that were approved and administered to people in 180 countries. LNPs are spheric drug delivery bodies which can be equipped with therapeutic payloads for intracellular delivery.

Intouch Solutions
SEPTEMBER 27, 2024
The discourse wasn’t just about cycling through tissue or aiding clinical development; it ranged from mutation prediction to tumor microenvironment (TME) analysis to how AI can aid in diagnosis and treatment planning to achieve better outcomes for patients. And that’s something new. And that’s, that’s a milestone.

Delveinsight
MARCH 2, 2021
To sum it, Chinook will manage clinical development and commercialization of product candidates while Evotec will be eligible for an undisclosed upfront payment, research funding, progress-dependent milestone payments, and tiered royalties on net sales for targets identified through the collaboration.

The Pharma Data
JUNE 28, 2021
It has been demonstrated to inflict difficult to repair damage to tumor cells by inducing DNA double strand breaks. Xofigo is currently under further evaluation in a broad clinical development program in prostate cancer and beyond. Compared to beta radiation, alpha radiation has a higher potency and shorter range.

The Pharma Data
OCTOBER 14, 2020
Priothera will use the funds to progress the clinical development of mocravimod, a modulator of sphingosine 1 phosphate (S1P) receptors, to enhance the curative potential of allogeneic hematopoietic stem cell transplantation (HSCT) for treating AML.

Roots Analysis
NOVEMBER 3, 2023
NAD + is involved in a wide range of cellular processes, including energy metabolism, DNA repair, and gene expression. Despite their potential therapeutic benefits, NAMPT inhibitors have faced several challenges in clinical development.

XTalks
OCTOBER 26, 2023
She has 23 years of experience working with clients to move vaccine candidates through the clinical development pipeline, including regulatory submission. The Flu versus COVID-19: Virology and Vaccines SARS-CoV-2 versus Influenza Virology Viruses contain genetic material that can either be RNA or DNA.

Advarra
AUGUST 10, 2023
Given the relatively small populations affected by any one rare disease or condition, a pharmaceutical company developing an orphan drug may reasonably expect the final approved drug to generate relatively small sales (when compared with the drug development costs) and consequently incur a financial loss.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content