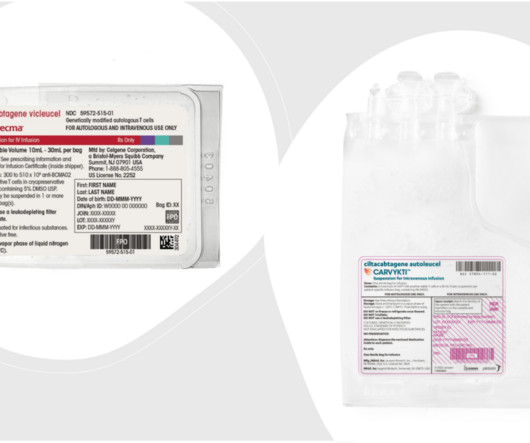
J&J’s Carvykti and BMS’ Abecma Win FDA Approvals for Earlier Use in Multiple Myeloma
XTalks
APRIL 11, 2024
Moreover, rather than producing antibodies, the cancer cells produce abnormal proteins that can cause complications. The companies have also expanded a contract manufacturing agreement with Novartis that will help produce Carvykti for both commercial and clinical supply at the New Jersey plant through the end of 2029.









Let's personalize your content