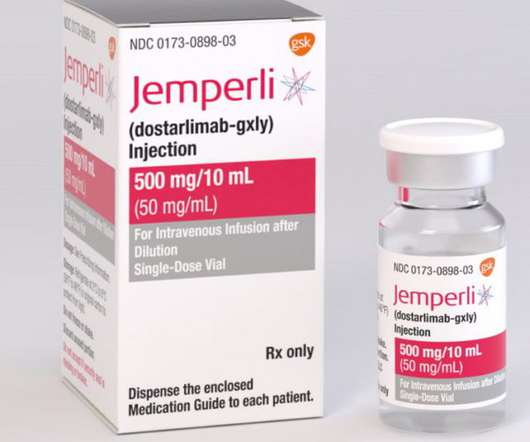
GSK receives FDA accelerated approval for JEMPERLI (dostarlimab-gxly) for adult patients with mismatch repair-deficient (dMMR) recurrent or advanced solid tumours
The Pharma Data
AUGUST 17, 2021
Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s). Mismatch repair-deficient tumours contain abnormalities that affect the proper repair of DNA when copied in a cell [i]. by blinded independent central review. with a DoR range of 2.6-35.8+












Let's personalize your content