FDA Warns About Compounded Versions of Ozempic and Wegovy
XTalks
JUNE 5, 2023
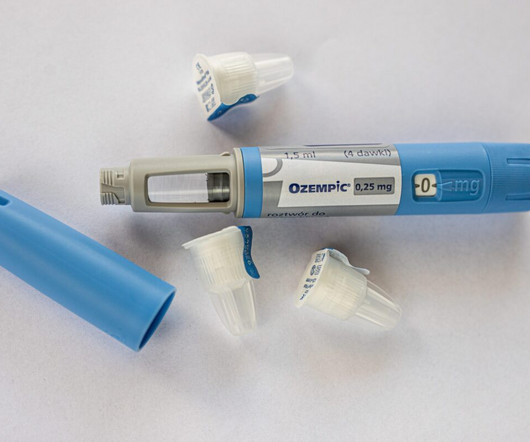
Ozempic and Wegovy, which contain the active ingredient semaglutide, were approved by the US Food and Drug Administration (FDA). Ozempic was approved for the management of type 2 diabetes mellitus in adults in addition to a reduced-calorie diet and exercise. The approved drugs contain the base form of semaglutide.













Let's personalize your content