T-cell vaccine for COVID-19 may last longer than current vaccines
The Pharma Data
APRIL 22, 2023
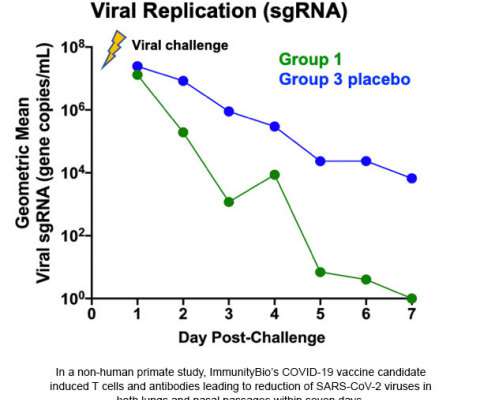
The current COVID-19 vaccines are designed to trigger an antibody response to the SARS-CoV-2 spike protein, which is vulnerable to mutations that could make the vaccine less effective over time. of the mice that were vaccinated with the T-cell-based vaccine survived, while only one of the control-group mice survived.













Let's personalize your content