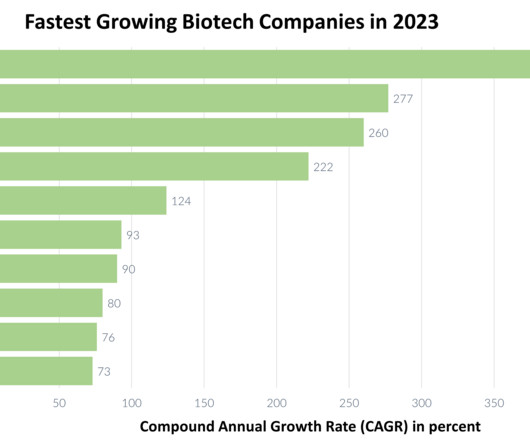
BioNTech’s RNA vaccine sparks potential in pancreatic cancer
Pharmaceutical Technology
MAY 11, 2023
Results published in Nature for a personalised pancreatic cancer vaccine that uses neoantigens from patients’ tumours have lent further support to early positive signals. The vaccine, developed by BioNTech, led to half of the patients with pancreatic cancer in the Phase I trial remaining cancer-free 18 months later.










































Let's personalize your content