Why small particles matter: Manufacturing and micro-material contamination
Pharmaceutical Technology
MARCH 6, 2024
To protect patient safety and reduce recall risks, drug manufacturers must reduce particle contamination sources wherever possible.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
MARCH 6, 2024
To protect patient safety and reduce recall risks, drug manufacturers must reduce particle contamination sources wherever possible.

Fierce Pharma
SEPTEMBER 25, 2023
GSK manufacturing review prompts Scynexis to pull Brexafemme, halt trials over contamination concerns esagonowsky Mon, 09/25/2023 - 12:26
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Bio Pharma Dive
APRIL 15, 2024
Because live cell and tissue products are extremely sensitive to contamination from microorganisms, viral particles or other airborne impurities, research and manufacturing involving these materials needs to be conducted in a cleanroom environment.

Fierce Pharma
JUNE 6, 2024
When the FDA comes knocking at your drug manufacturing facility, it’s best to play along. |

Bio Pharma Dive
APRIL 22, 2021
A recent nine-day inspection by agency officials found numerous breaches of manufacturing standards and inadequate training for factory employees.

XTalks
JULY 16, 2021
Johnson & Johnson is recalling sunscreens from two of its popular brands over concerns of carcinogenic benzene contamination. Therefore, levels should be monitored at different stages of production to avoid harmful benzene contamination. Growing Contamination Issues. Despite this, contamination issues still persist.

Fierce Pharma
APRIL 10, 2024
FDA chided Kilitch Healthcare India for “poor practices” tied to written procedures around microbial contamination, shoddy lab records, behaviors that could have caused contamination and quality control lapses. . | In a four-observation warning letter issued this week, the U.S.
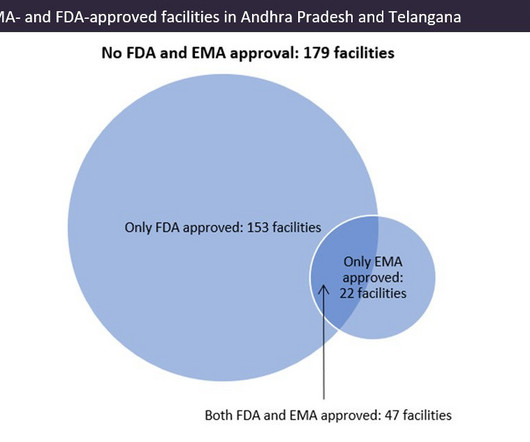
Pharmaceutical Technology
APRIL 4, 2023
Indian pharma manufacturing continues to be the backbone of drug supplies worldwide, and GlobalData analysis suggests US overreliance on the country for generic drug supply. Pharma manufacturing facilities in Andhra Pradesh and Telangana accounted for 22.5% © GlobalData. © GlobalData.
STAT News
AUGUST 22, 2022
After a decade of manufacturing problems, a U.S. federal court ordered a unit of Wockhardt, one of the largest makers of generic drugs, to refrain from making allegedly adulterated medicines at a facility in Illinois. However, the U.S. Continue to STAT+ to read the full story…
XTalks
MAY 19, 2022
According to the company, products were manufactured by a third party and subsequently distributed across North America. Since metal contamination is a common issue in food production, resulting in expensive recalls and lawsuits, the FDA has implemented metal detection standards to ensure the same principles are followed for all companies.

Pharmaceutical Technology
JULY 14, 2022
Regulatory decisions have a ripple effect, first affecting the pharma sponsors, and then the companies tasked to manufacture the drug. Pharmaceutical Technology looks at drugs and biologics with recent regulatory verdicts that will likely impact manufacturing volumes. Consequential FDA and EMA decisions.

BioPharma Reporter
OCTOBER 6, 2021
A probe has identified 'human errorâ at the Spanish contract development and manufacturing organization, Rovi, as the reason for the presence of metal contaminants in Moderna COVID-19 vaccine doses, leading to Japanese authorities withdrawing 1.6 million doses of the shot in August.

Pharmaceutical Technology
OCTOBER 26, 2022
Nano-based delivery systems are on the rise, as they enable manufacturers to deliver therapeutic agents to specific targeted tissue in a more controlled manner. Regulations and guidelines for nanopharmaceuticals are still relatively in their infancy, including for cleaning processes and the prevention of cross-contamination.

STAT News
APRIL 3, 2023
Your tips and insights help the world go around… Global Pharma Healthcare, whose eye drops have been linked to dozens of serious reactions, failed to follow numerous procedures to ensure its products did not become contaminated , according to a U.S. Food and Drug Administration inspection report obtained by STAT.

XTalks
MAY 18, 2023
Cornell University researchers have been making strides in the development of a revolutionary tool to combat dangerous pathogens in commercial dry food manufacturing plants. Food manufacturers use steam for tasks like dehydration, distillation, pasteurization and cooking, which helps preserve the taste and texture of their offerings.

Roots Analysis
APRIL 15, 2024
Further, the manufacturing of biologics fill finish is a highly complex and cost-intensive process. Notable examples of fill finish manufacturing companies equipped with SA25 aseptic filling workstation include ( in alphabetical order ) Emergent Bioservices, PCI Pharma Services, Singota Solutions and WuXi Biologics.

pharmaphorum
MARCH 22, 2022
In this article, Ben Hargreaves asks whether there is particular potential for the technologies to revolutionise staff training and reduce costs in pharma manufacturing. The importance of manufacturing. the streamlining of pharma manufacturing in the 21st century. Taking manufacturing to the next level.

BioSpace
APRIL 29, 2021
Battered by vaccine manufacturing mishaps that led to the contamination of 15 million doses of the Johnson & Johnson vaccine, Emergent BioSolutions is shaking up its leadership to ensure such errors do not occur again.

XTalks
AUGUST 12, 2024
Lead, a toxic metal, can enter the food supply through environmental contamination during the growing, raising or processing of foods. FDA’s Regulatory Framework on Lead in Food Lead contamination in food can arise from various environmental sources, including past usage of lead in paint, gasoline and plumbing materials.

Fierce Pharma
FEBRUARY 20, 2024
It’s been 15 years since Genzyme began rationing the Fabry disease treatment Fabrazyme after a shortage caused by contamination at a manufacturing site, marking the start of many years of litigatio | Sanofi's Genzyme was accused of causing injuries after contaminated doses of its Fabry disease drug led to a shortage, during which the company rationed (..)

The Pharma Data
MARCH 29, 2022
an animal food manufacturing company of Carney’s Point, New Jersey, has agreed to stop selling, manufacturing and distributing raw pet food and come into compliance with the Federal Food, Drug, and Cosmetic Act (FD&C Act). Food and Drug Administration (FDA) announced today that Bravo Packing, Inc.,

XTalks
JULY 30, 2024
The US Department of Agriculture’s (USDA) Food Safety and Inspection Service (FSIS) declared Salmonella an adulterant in raw breaded stuffed chicken products when contamination levels exceed one colony-forming unit (CFU) per gram. This approach emphasizes stringent testing and preventive measures at earlier stages of the supply chain.

Pharmaceutical Technology
AUGUST 17, 2022
NTI have tackled this problem through the introduction of its NEBULAE® SRS system , which filters and recycles the air in surgical theatres, removing potentially harmful contaminates. has been manufacturing, designing, and innovating since 1984. Pioneers in minimally invasive surgical technology, Northgate Technologies Inc.

Medical Xpress
JANUARY 4, 2023
The researchers also determined the relative contribution of various sources of antibiotic contamination in waterways, such as hospitals, municipals, livestock, and pharmaceutical manufacturing.

Medical Xpress
APRIL 17, 2023
An analysis of global usage patterns of a common hypertension drug following a major recall demonstrates the worldwide impact of contamination at a single manufacturing facility.

STAT News
JANUARY 19, 2023
In a stunning rebuke, the Food and Drug Administration accused a drugmaker of a “cascade of failures” for a litany of quality-control problems at a manufacturing plant, the latest instance in which the regulator has castigated an Indian pharmaceutical company for such lapses. Continue to STAT+ to read the full story…

Roots Analysis
SEPTEMBER 20, 2023
The focus of stakeholders has now shifted to optimizing the cell therapy manufacturing process. It is worth noting that manufacturing process biologics and cell therapies is considerably complex when compared to small molecule drugs. The key steps involved in the supply chain of cell therapy manufacturing are presented below.

STAT News
MARCH 31, 2023
Global Pharma Healthcare, whose eye drops have been linked to dozens of serious reactions, failed to follow numerous procedures to ensure its products did not become contaminated, according to a Food and Drug Administration inspection report obtained by STAT.

pharmaphorum
JANUARY 30, 2023
Light is being shed on recent product recalls for mould contamination, and infection outbreaks serve as a reminder to pay more attention to these overlooked organisms, as well as considerations towards container closure integrity testing which is being increasingly deployed to block microbe movement.

Roots Analysis
OCTOBER 24, 2023
The limitations of stainless-steel bioreactors such as high risk of contamination, process related challenges and high operational cost have prompted innovators to develop more advanced single use bioreactor in order to meet the rising demand for safe and efficacious biotherapeutics.

STAT News
JANUARY 10, 2023
Food and Drug Administration for a host of serious manufacturing violations at a key plant in India, the latest instance in which the company was tagged by the regulator for quality-control problems.

Roots Analysis
JANUARY 12, 2023
The focus of stakeholders has now shifted to optimizing the cell therapies manufacturing process. For the success of cell-based therapies , an effective manufacturing platform and a robust supply chain model is imperative. The key steps involved in the supply chain of cell therapies manufacturing are presented below.
XTalks
APRIL 17, 2023
The causes of these events varied, from contaminated food and water to zoonotic diseases and poor hygiene practices. It was caused by contaminated ice cream products manufactured by Blue Bell Creameries, a popular ice cream company based in Texas. Related: Here’s Why Raw Flour is Linked to Foodborne Illness Outbreaks 1.
XTalks
NOVEMBER 23, 2023
Salmonella Outbreak from Poultry — The US A widespread Salmonella outbreak linked to contaminated poultry affected thousands across multiple states this year. It also encouraged a citizen petition to make it illegal to sell poultry contaminated with any one of 31 strains of Salmonella. Coli Daycare Outbreak — Canada In September, an E.

Medical Xpress
FEBRUARY 16, 2023
Like any other manufactured food product, chocolate can be contaminated if key ingredients or processes break down.

pharmaphorum
MAY 5, 2022
Assuring end-patient safety through holistic contamination control. Dissect the framework of a robust Global Contamination Control Program and how to approach implementing a holistic CCS. Workshop Leaders: Ziva Abraham, President, Microrite and Morgan Polen, SME Cleanroom Contamination Control, Microrite.

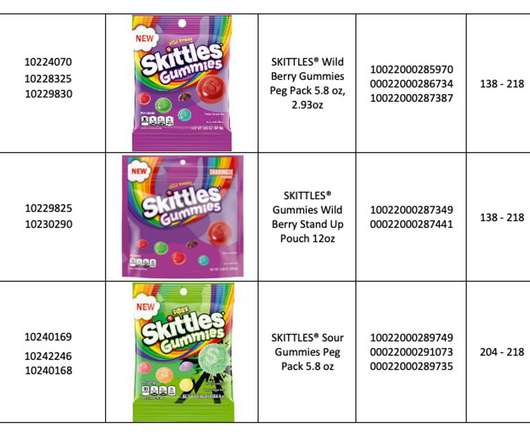
XTalks
MAY 26, 2022
In this episode of the Xtalks Food Podcast, Sydney talks about the latest Mars recall, which included Starburst, Skittles and Life Savers gummies that were manufactured by a third party and distributed across North America. Kidfresh: The Better-For-You Kid’s Brand with Hidden Veggies.

XTalks
AUGUST 15, 2024
These tiny particles, primarily from environmental contamination, can infiltrate the food supply. In this episode of the Xtalks Food Podcast, Sydney talks about how microplastics and nanoplastics primarily contaminate food through environmental exposure where crops and livestock are raised.

Drug Discovery World
JULY 11, 2024
Download this interactive infographic to: Discover ways to navigate your contamination, compliance, reproducibility, monitoring and scalability challenges Identify which critical factors and equipment you should consider to help succeed in a GMP environment Unlock access to an extensive collection of helpful resources Download now.

XTalks
AUGUST 7, 2023
coli outbreak between 1992 and 1993 that claimed the lives of four children who had eaten contaminated burgers from the fast-food chain Jack in the Box. The spotlight frequently fell on corporate executives and government regulators in the Netflix food documentary, these being the usual suspects behind food contamination.
XTalks
MARCH 25, 2022
Pfizer places the utmost emphasis on patient safety and product quality at every step in the manufacturing and supply chain process,” the company stated in a news release. Last summer, Pfizer recalled a dozen lots of its smoking cessation drug Chantix due to nitrosamine contaminants.

XTalks
MAY 3, 2024
Approximately one in every 25 chicken packages at grocery stores is contaminated with Salmonella. Other food products can also be contaminated with Salmonella. coli , initiated after deadly outbreaks linked to contaminated ground beef, which resulted in a reduction of related illnesses by more than 50 percent.

STAT News
AUGUST 15, 2022
communities. Although researchers have known about the impact of these chemicals for years, there has been little guidance available for clinicians.

NY Times
MAY 10, 2022
The report sheds new light on executives’ worries about deficiencies in the company’s quality control systems at its troubled Baltimore plant; no contaminated doses were ever released to the public.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content