Positive in vitro results for Imutex’s FLU-v
Pharma Times
OCTOBER 1, 2022
Data further supports the continued development of FLU-v as a broad-spectrum influenza vaccine
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

Pharma Times
OCTOBER 1, 2022
Data further supports the continued development of FLU-v as a broad-spectrum influenza vaccine

World of DTC Marketing
JULY 12, 2021
SUMMARY: Pfizer says it has data on waning immunity from their COVID vaccine. According to a report on CNN.com “Pfizer said it is seeing waning immunity from its coronavirus vaccine and says it is picking up its efforts to develop a booster dose that will protect people from variants.” ” So who to believe?
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

BioPharma Reporter
JANUARY 25, 2021
An in vitro study shows Moderna's existing mRNA COVID-19 vaccine provides protection against strains that have emerged from South Africa and the UK. But it does suggest reduced protection against the South Africa strain, and so the company is also developing a new booster designed for maximum efficacy against this variant.

BioPharma Reporter
JANUARY 28, 2021
In vitro studies found the Pfizer/BioNTech COVID-19 vaccine elicits antibodies against both the UK and South African virus strains.

XTalks
MARCH 1, 2021
(NYSE: PFE ) and BioNTech SE (Nasdaq: BNTX ) have announced that they are beginning a trial to evaluate the safety and efficacy of a third booster dose for their COVID-19 vaccine (BNT162b2), as well as new vaccine variants. Related: Could Pfizer and BioNTech’s COVID-19 Vaccine be Stored at Standard Freezer Temperatures?
pharmaphorum
MARCH 4, 2021
UK chancellor Rishi Sunak has announced a budget loaded with initiatives designed to kick-start the UK’s economy as it recovers from the coronavirus pandemic, with vaccine development, pharma and life sciences playing a key role. A scheme providing £500 payments to people self-isolating has been extended in England until the summer.

Camargo
NOVEMBER 29, 2021
The development of biological products (or biologics) represents a major advancement in modern medicine, enabling the treatment of patients with many illnesses where no other therapeutics were previously available. Biologics include a wide range of products , including: Vaccines. Definition of Biologic Products. Allergenics.

Pharmaceutical Technology
MAY 12, 2023
BioNTech has ended its research collaboration with Matinas after its oral mRNA vaccine failed to demonstrate preclinical activity. Matinas said it developed this unique formulation to “handle the physical complexity and biological fragility of mRNA”. Matinas announced the news in a May 10 statement.
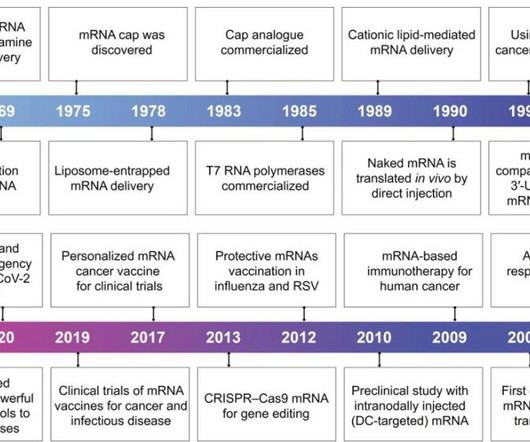
Pharma Mirror
APRIL 21, 2024
With the rapid development of biotechnology and molecular medicine, the introduction of mRNA as a vaccine or therapeutic agent enables the production of almost any desired functional protein/peptide within the human body.

XTalks
JANUARY 15, 2021
Ortho Clinical Diagnostics received emergency use authorization (EUA) from the US Food and Drug Administration (FDA) for its VITROS SARS-CoV-2 antigen test for detection of active COVID-19 infection. Related: COVID-19 Vaccine and Drug Development Coverage. active virus).
Pharmaceutical Technology
NOVEMBER 22, 2022
Vaccines are our number one weapon in the fight against infectious diseases, but their development has historically involved a long and complex process taking up to a decade. Before COVID-19, Merck held the record for the fastest modern vaccine ever developed. mRNA’s potential for rapid vaccine delivery.
Pharmaceutical Technology
JUNE 1, 2023
Pneumagen has raised £8m ($10 million) for the further development of its intranasal antiviral drug neumifil in Phase II studies for chronic obstructive pulmonary disease (COPD) patients suffering from virus-induced exacerbations. The vaccine is self-administered through an intranasal spray, and is based on the company’s GlycoTarge platform.

XTalks
JUNE 27, 2022
Leading global medical technology company Becton Dickinson (BD) and Spain’s CerTest Biotec have announced they will be collaborating to develop a molecular diagnostic test for the detection of the monkeypox virus. There is currently no commercial test available for the detection of the monkeypox virus.
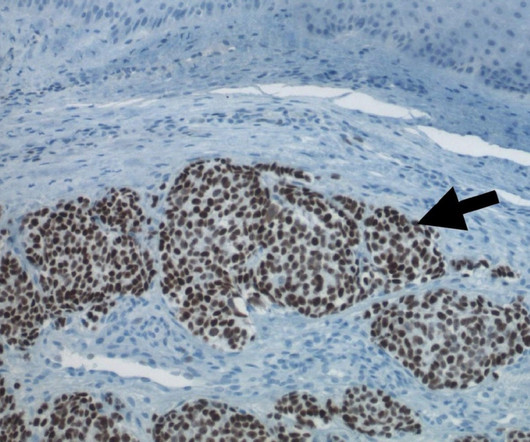
Pharmaceutical Technology
MAY 24, 2023
Our board and management team believe that the combined company will be well-positioned to develop powerful new therapies with the potential to overcome resistance to current immunotherapies, an area of significant unmet need.” Morphogenesis’ lead personalised cancer vaccine, IFx-Hu2.0,

pharmaphorum
AUGUST 4, 2022
Training and development. Staff development and training programmes were largely on pause during the pandemic. Businesses need to ensure they prioritise the recruitment and development of staff to help ensure longevity and survival. Harness technology.

pharmaphorum
JANUARY 22, 2021
So far, neither variant seems to be associated with more severe COVID-19 symptoms, although there has been some preliminary research suggesting the SA strain could allow reinfection with SARS-CoV-2, and also be less susceptible to vaccines. The UK strain – known as B.1.1.7

pharmaphorum
OCTOBER 18, 2021
The development of COVID-19 vaccines required levels of cooperation and pioneering science comparable to the Apollo space programme. Within 12 months of the outbreak, vaccines were being deployed to prevent severe infections, hospitalisation, and death. billion people have been fully vaccinated. And there are many.
XTalks
NOVEMBER 15, 2023
France-based biotech Valneva has won approval from the US Food and Drug Administration (FDA) for its chikungunya vaccine Ixchiq for the prevention of infection from the chikungunya virus. The vaccine is approved for adults 18 years of age and older who are at increased risk of exposure to the virus.

XTalks
MARCH 11, 2021
Amidst initial confusion, fear and chaos, masks and social distancing quickly became new norms, and now vaccines are leading hope for a way out. With vaccine shortages and slow rollouts in many parts of the world, the COVID-19 way of life continues in many places. As of March 11, 2021, COVID-19 has claimed the lives of over 2.6

XTalks
APRIL 27, 2022
In the US, the vaccination for the virus is currently provided to individuals five years of age and older. The in vitro studies showed that remdesivir was effective among the drugs because it had the strongest antiviral activity as it could block virus infection at low micromolar concentrations with minimal cell toxicity.

Pharmaceutical Technology
SEPTEMBER 10, 2008
The first activities of the company concerned the pre-clinical toxicological testing of several new candidate molecules showing cytostatic activity, initially developed by a former research institute, as well as the breeding and supplying of laboratory animals. . It will most probably lead to a new anti-flu vaccine.

pharmaphorum
DECEMBER 16, 2020
We had companies collaborating to develop laboratories. Soon the ABPI and the entire industry were facing the ripple effects of COVID-19, including a significant slowdown in non-COVID clinical research, which Torbett says was challenging for the industry to deal with. “We’ve We had companies sourcing consumables for testing machines.
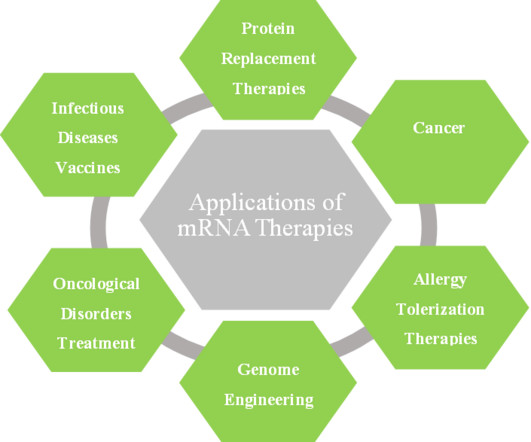
Roots Analysis
MARCH 16, 2023
In the last few years, researchers have become interested in using in vitro transcribed (IVT) mRNA as a drug delivery agent. It is worth noting that several companies have begun to develop mRNA-based cancer immunotherapies and vaccines for infectious diseases.
XTalks
JANUARY 27, 2021
COVID-19 vaccine maker Moderna announced this week that results from a study show that the shot is effective against some of the new circulating variants of SARS-CoV-2. In a news release from Moderna, the company reported that the administration of its COVID-19 vaccine induced antibody titers against both variants.
The Pharma Data
AUGUST 15, 2021
Even as several safe and effective COVID-19 vaccines are being administered to people worldwide, scientists are still hard at work developing different vaccine strategies that could provide even stronger or longer-lasting immunity against SARS-CoV-2 and its variants.
pharmaphorum
OCTOBER 5, 2022
The US Government’s Biomedical Advanced Research and Development Authority (BARDA) has made an initial investment of approximately $55 million for rapid development of VIR-2482, the Vir Biotechnology’s investigational prophylactic monoclonal antibody (mAb) for seasonal and pandemic influenza viruses.

Pfizer
JANUARY 27, 2023
In the ongoing development of the Pfizer-BioNTech COVID-19 vaccine, Pfizer has not conducted gain of function or directed evolution research. This research provides a way for us to rapidly assess the ability of an existing vaccine to induce antibodies that neutralize a newly identified variant of concern. and around the world.
The Pharma Data
MARCH 11, 2021
(NYSE: PFE) and BioNTech SE (Nasdaq: BNTX) today announced real-world evidence demonstrating dramatically lower incidence rates of COVID-19 disease in individuals fully vaccinated with the Pfizer-BioNTech COVID-19 Vaccine (BNT162b2), underscoring the observed substantial public health impact of Israel’s nationwide immunization program.

pharmaphorum
JANUARY 17, 2023
Research into mRNA dates back to the 1970s, but with the approval of both Moderna and BioNTech/Pfizer’s vaccines, this approach has finally been validated. At the end of last year, the UK government entered into a partnership with the company, which saw Moderna agree to provide 250 million vaccine doses per year over the course of a decade.

The Pharma Data
SEPTEMBER 17, 2020
This is the focus of vaccines in development and convalescent plasma therapy. The majority of current candidate vaccines aim to induce an antibody response against the spike protein. “As The test targets antibodies against the spike protein. Food and Drug Administration (FDA). said Thomas Schinecker, CEO Roche Diagnostics.

XTalks
MARCH 27, 2024
In vitro diagnostics, or IVD, is a field where precision, reliability and robust partnerships propel advancement. Each diagnostic evaluation and every lab discovery signify not only technological advancement but also the fruit of integrated teamwork, with decisions made collaboratively. Their significance in the field of IVD is paramount.

The Pharma Data
MAY 17, 2021
The European Commission’s Medical Device Coordination Group (MDCG) sent a notice to in vitro diagnostic (IVD) makers and their authorized representatives reminding them of their obligation to assess the impact of COVID-19 variants on the performance of their tests. . Posted 17 May 2021 | By Michael Mezher .

Pharmaceutical Technology
AUGUST 26, 2008
The dynamic has now changed as many big companies have now acquired biotechnology companies or have developed ‘biotechnology divisions’ of their own. It was once derided as the stuff of science fiction, but in recent years, biotechnology has emerged as an important growth area in pharmaceuticals.
pharmaphorum
NOVEMBER 4, 2020
However, in vitro or binding-based efficacy insight needs to be confirmed by reliable clinical evidence, as shown by the outcomes of COVID-19 trials with remdesivir and hydroxychloroquine. million worldwide and continues to increase despite the global public health measures that have been put in place.

The Pharma Data
JANUARY 11, 2021
a San Diego-based biotechnology company with an array of technology platforms for antibody discovery and optimization, and novel NK and T cell engager generation, today announced licensing of a panel of its anti-SARS-CoV-2 antibody clones to IGM Biosciences for COVID-19 therapy development.

pharmaphorum
JANUARY 16, 2023
There were developments in the past 12 months, true – Biogen/Eisai’s lecanemab (now branded Leqembi) showed clear success in the Clarity AD trial and received approval by FDA on 6th January 2023. The hard problem: innovation for high prevalence, chronic diseases of ageing.

Delveinsight
JANUARY 6, 2021
As a result of the high healthcare cost, people from developed countries move from their home country to another to receive treatment. As a result of the high healthcare cost, people from developed countries move from their home country to another to receive treatment. What is Medical Tourism?

XTalks
SEPTEMBER 30, 2020
The researchers thought there may be similar mutations among COVID-19 patients, and that they could identify individuals at risk of developing severe disease as well as provide leads for new therapies, explained Casanova. Related: Low T Cell Counts Observed in COVID-19 Patients. Mutational Errors in COVID-19 Patients. percent of women and 12.5

The Pharma Data
NOVEMBER 17, 2021
Streamlined in vitro data, published in bioRxiv, demonstrate that sotrovimab retains exertion against all current variants of concern and interest of the SARS-CoV-2 contagion as defined by the World Health Organization, plus others, including, but not limited to, Delta (B.1.617.2), 1.617.2), Delta Plus (AY.1 2) and Mu (B.1.621).

Pfizer
AUGUST 15, 2022
I am grateful to have received four doses of the Pfizer-BioNTech vaccine and I am feeling well while experiencing very mild symptoms. I am incredibly grateful for the tireless efforts of my Pfizer colleagues who worked to make vaccines and treatments available for me and people around the world.

Pfizer
DECEMBER 13, 2022
It was developed to be administered orally so that it can be prescribed early after infection, potentially helping patients avoid severe illness (which can lead to hospitalization and death). Nirmatrelvir has shown consistent in vitro antiviral activity against the following variants: Alpha, Beta, Delta, Gamma, Lambda, Mu, and Omicron BA.1,
Delveinsight
FEBRUARY 23, 2021
The company develops transformative biologics including vaccines and therapeutics for the world’s most debilitating diseases. The company plans to use the capital to expand its pipeline of protein-based vaccines and biologic cancer therapies utilizing its innovative and proprietary Trimer-Tag technology platform.

The Pharma Data
MARCH 23, 2021
The oral antiviral clinical candidate PF-07321332, a SARS-CoV2-3CL protease inhibitor , has demonstrated potent in vitro anti-viral activity against SARS-CoV-2, as well as activity against other coronaviruses, suggesting potential for use in the treatment of COVID-19 as well as potential use to address future coronavirus threats.
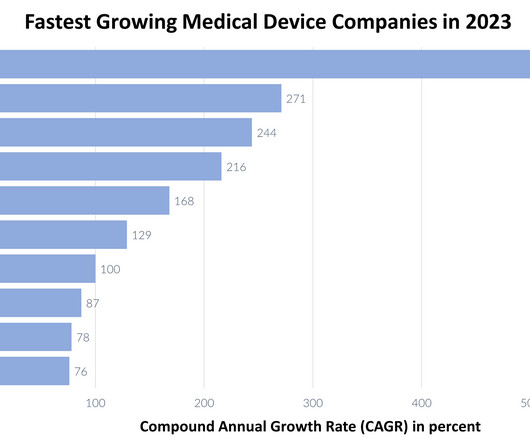
XTalks
DECEMBER 11, 2023
Through the utilization of their state-of-the-art technology and substantial investments in research and development (R&D), these med tech/medical device companies persist in driving industry innovation beyond the pandemic, thereby laying the foundation for a more streamlined approach against diseases in the future.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content