Egyptian Drug Authority approves Lilly and EVA’s insulin injection
Pharmaceutical Technology
DECEMBER 18, 2024
Eli Lilly and EVA Pharma have announced approval from the Egyptian Drug Authority for the human insulin glargine injection.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

Pharmaceutical Technology
DECEMBER 18, 2024
Eli Lilly and EVA Pharma have announced approval from the Egyptian Drug Authority for the human insulin glargine injection.

Pharmaceutical Technology
JULY 15, 2022
Insulin prices made the headlines again as California governor Gavin Newsom announced plans on 7 July for the state to manufacture low-cost insulin. The state plans to work directly with a contract manufacturing organization (CMO) to manufacture low-cost insulin. In-house manufacturing the norm.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

World of DTC Marketing
MARCH 31, 2022
The House is preparing to vote on a $35 monthly insulin cap later, but there is stern opposition to the plan, which could cost billions over ten years. Essentially taxpayers would be subsidizing drug company profits for the price of a product that should have come down a long time ago. This could cost taxpayers billions over the year.
Bio Pharma Dive
MARCH 3, 2022
The nonprofit group announced ambitious plans to begin offering insulin at prices no higher than $30 per vial by early 2024.

Bio Pharma Dive
MAY 16, 2024
The results, which showed Lilly’s drug was as effective at controlling blood sugar as standard injections, come as Novo Nordisk seeks approval for a competing once-weekly insulin.

Bio Pharma Dive
MARCH 1, 2023
The pharma company, along with its diabetes drug rivals Novo Nordisk and Sanofi, has long been under pressure over the high cost of insulin — scrutiny that has recently ramped up in the U.S.

Bio Pharma Dive
MARCH 14, 2023
Responding to policy changes and pressure over high drug costs, the pharma is reducing the sticker price of four branded diabetes medicines by up to 75%.

Pharmaceutical Technology
OCTOBER 12, 2022
Novo Nordisk has recently completed its six-part ONWARDS Phase III trial, as ONWARDS 5 reached its primary endpoint with Icodec demonstrating non-inferiority in reducing hemoglobin A1C (HbA1c) in patients with type 2 diabetes (T2D) at week 52 in comparison to once-daily basal insulin analogs. Patients had an overall baseline HbA1c of 8.9%

Bio Pharma Dive
NOVEMBER 18, 2022
Eli Lilly’s long-acting copycat drug, first approved in late 2021, now has a designation that will allow pharmacists to swap it for Sanofi’s Lantus.

AuroBlog - Aurous Healthcare Clinical Trials blog
JULY 10, 2024
A class of diabetes medications, which include the best-selling drug Ozempic, are associated with a reduced risk of certain obesity-related cancers, according to a study released Friday.

Bio Pharma Dive
MARCH 22, 2023
The companies will collaborate to identify compounds that could treat insulin resistance by targeting shape-shifting cellular droplets known as biomolecular condensates.

pharmaphorum
APRIL 13, 2022
The treatment has been around for over a century but insulin has still become the centre of the ongoing discussion over pricing of pharmaceutical products. Ben Hargreaves looks at why insulin’s price is so controversial and whether this could be set to change. Insulin rationing. In the US, approximately 37.3

XTalks
FEBRUARY 18, 2025
The FDA has approved Sanofis Merilog (insulin-aspart-szjj) as the first rapid-acting insulin biosimilar product for the treatment of diabetes. Merilog is a biosimilar to Novo Nordisks NovoLog, a widely used rapid-acting insulin aspart. Insulin, a hormone produced by the pancreas, allows glucose to enter cells for energy.

Bio Pharma Dive
JULY 29, 2021
An injectable insulin from Viatris has become the first-ever biosimilar product that can be directly substituted for a marketed biologic, a long-awaited decision that could put pricing pressure on other diabetes drugs.
Bio Pharma Dive
MARCH 27, 2023
Vertex will license CRISPR technology to develop insulin-producing islet cells that are more resistant to immune rejection, adding to other efforts directed at Type 1 diabetes.
Pharmaceutical Technology
MAY 26, 2023
Chronic disease tech company Convatec Group has partnered with Beta Bionics to manufacture the insulin delivery system iLet Bionic Pancrease. According to Beta Bionics, this is the only first and only automated insulin delivery system that carries 100% of doses and does not require carb counting.

Bio Pharma Dive
JULY 14, 2021
Rival Novo Nordisk has jumped ahead in developing an injectable drug that responds to changes in blood sugar. An acquisition of Protomer Therapeutics could help Lilly close the gap.

Bio Pharma Dive
FEBRUARY 8, 2023
In a State of the Union address, Biden threatened to veto any efforts to repeal the legislation and chastised drugmakers for the current costs of insulin.

pharmaphorum
MAY 1, 2024
Polish drug delivery company Biotts says it has demonstrated delivery of insulin across the skin for the first time without the use of an injection system.

Bio Pharma Dive
JULY 10, 2024
Antitrust regulators are set to file suit against CVS Caremark, Express Scripts and Optum Rx over how they negotiate discounts for drugs, including insulin, per the report.

XTalks
OCTOBER 8, 2021
Medtronic has expanded a Class I recall of remote controllers used with the MiniMed 508 insulin pump or the MiniMed Paradigm family of insulin pumps over potential cybersecurity risks. The medtech giant has also expanded another Class I recall of the retainer rings on its 600 series insulin pumps.

pharmaphorum
JULY 29, 2021
Generic drugmaker Mylan has become the first company to secure FDA approval for a biosimilar product that is considered completely interchangeable with the reference product – namely Sanofi’s once-daily insulin Lantus.

pharmaphorum
JANUARY 13, 2023
The ‘big three’ insulin producers Novo Nordisk, Eli Lilly, and Sanofi are being sued by the state of California for allegedly working together to set artificially inflated prices for their products. The suit follows legal action over insulin pricing taken by other states, including Arkansas , Kansas, Mississippi, and Minnesota.
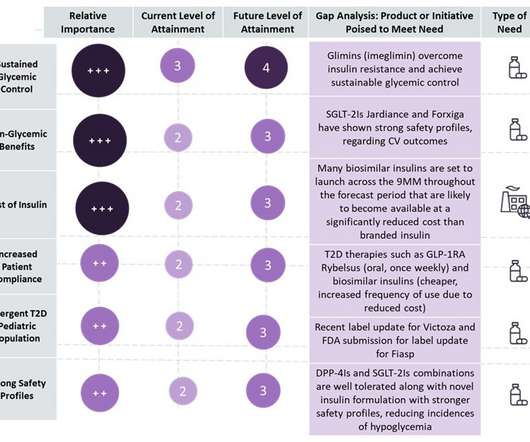
Pharmaceutical Technology
JANUARY 10, 2023
Type 2 diabetes (T2D) is a crowded and competitive landscape with multiple “me-too,” generic and biosimilar drugs entering the market, with market growth primarily driven by an increasing prevalent population across nine major markets (9MM: US, France, Germany, Italy, Spain, UK, Japan, China, and India).

Medical Xpress
MARCH 17, 2023
French pharmaceutical giant Sanofi announced on Thursday it would cut the US price of its most prescribed insulin by 78 percent, falling in line with two other major drug makers.

pharmaphorum
APRIL 29, 2022
For many patients with type 2 diabetes, having to inject basal insulin every day is a burdensome fact of life, but Novo Nordisk is trying to change that. cases of hypoglycaemia per year with insulin icodec, versus 0.27 events for the once-daily drug. In absolute terms, there were 0.73
STAT News
JANUARY 20, 2023
In an attempt to tackle the root causes of inflated insulin prices, the state of California filed a lawsuit last week accusing drug manufacturers and pharmacy benefit managers of artificially and illegally jacking up the price of insulin. The sausage-making of how insulin is priced is not for the faint of heart.

NPR Health - Shots
SEPTEMBER 12, 2022
Drug price reforms passed as part of the Inflation Reduction Act did not solve the problem of skyrocketing costs of insulin. States are taking their own action.

XTalks
OCTOBER 28, 2024
Diabetes, a chronic condition marked by high blood glucose levels due to insulin issues, brings complications like heart disease, stroke, neuropathy and diabetic foot ulcers. Abbot also joined Medtronic to pair its sensors with the latter’s automated insulin delivery (AID) systems. million undiagnosed and 115.9

pharmaphorum
JULY 14, 2021
Eli Lilly bought a stake in Protomer Technologies and its glucose-sensing insulin platform last year, and it must like what it has seen in the programme since then – it has just agreed to buy the company outright. . It was also responsible for the first recombinant insulin product – Humulin – which launched in 1982 and remains on sale.

BioSpace
DECEMBER 28, 2020
rBIO launched last week with technology that can reduce the cost of insulin by 30 percent, making U.S. manufacturing cost-effective for insulin and several other drugs.

STAT News
DECEMBER 15, 2022
In a bid to widen access to insulin in primarily low-income nations in Africa, Eli Lilly has agreed to sell active pharmaceutical ingredients and transfer technology to an Egyptian company that expects to supply the life-saving diabetes treatment to as many as 1 million people by 2030.

BioSpace
MAY 27, 2024
Flagging a risk of hypoglycemia, the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee on Friday did not recommend the approval of Novo Nordisk’s once-weekly insulin icodec for type 1 diabetes.

Medical Xpress
NOVEMBER 15, 2022
In a new 'Ideas and Opinions,' authors from Johns Hopkins Bloomberg School of Public Health analyze California's newly announced effort to manufacture insulin for its residents. The commentary is published in Annals of Internal Medicine.

Drug Channels
SEPTEMBER 15, 2023
Nicole summarizes MMIT’s research on payers’ views of two crucial market changes: (1) the Inflation Reduction Act of 2022’s requirement to cap insulin prices at $35 for Medicare beneficiaries, and (2) manufacturers’ reduction in list prices for certain insulin products. d/b/a Drug Channels Institute.

BioSpace
MAY 22, 2024
In advance of Friday’s Endocrinologic and Metabolic Drugs Advisory Committee meeting, the FDA has raised concerns about hypoglycemia linked to Novo Nordisk’s insulin icodec, according to a briefing document.

Fierce Pharma
AUGUST 12, 2024
Despite hitting a roadblock on the path to approval for its once-weekly insulin icodec in the U.S.,

Medical Xpress
FEBRUARY 16, 2023
Two classes of drugs prescribed off-label for some patients with type 1 diabetes can provide significant benefits, but also come with health concerns, according to a study by UT Southwestern Medical Center researchers.

NPR Health - Shots
MARCH 1, 2023
The moves announced Wednesday promise critical relief to some people with diabetes who can face annual costs of more than $1,000 for insulin they need in order to live. Image credit: Darron Cummings/AP)

Bio Pharma Dive
SEPTEMBER 14, 2023
New weight loss drugs have sparked a stock selloff for some device makers, but medtech analysts don’t see them a likely to undermine the companies’ business — yet.

STAT News
JULY 15, 2022
Next year, UnitedHealthcare will offer employer health plans that have no copays or out-of-pocket costs for five vital drugs — insulin, epinephrine, glucagon, naloxone, and albuterol — but those discounts will only be a guaranteed for less than a quarter of UnitedHealthcare’s membership for now.

Pharmaceutical Technology
FEBRUARY 24, 2023
According to GlobalData, Phase III drugs for Type 2 Diabetes have a 53% phase transition success rate (PTSR) indication benchmark for progressing into Pre-Registration. GlobalData’s report assesses how (LAI-287 + semaglutide)’s drug-specific PTSR and Likelihood of Approval (LoA) scores compare to the indication benchmarks.

BioPharma Reporter
NOVEMBER 21, 2022
Oramed Pharmaceuticals, a company focused on the development of oral drug delivery platforms, has signed an exclusive commercial distribution agreement for the Republic of Korea with local player, Medicox Co Ltd, in relation to its oral insulin candidate.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content