Cell, gene therapy makers lose a champion at FDA with exit of Peter Marks
Bio Pharma Dive
MARCH 31, 2025
Marks’ resignation leaves the field without a regulator many view as “integral” to its progress over the last decade.

Bio Pharma Dive
MARCH 31, 2025
Marks’ resignation leaves the field without a regulator many view as “integral” to its progress over the last decade.

Pharmaceutical Technology
MARCH 31, 2025
The funding is set to accelerate the development of Isomorphics AI drug design engine and advance clinical programmes.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Bio Pharma Dive
MARCH 31, 2025
Shares in Moderna, Vaxcyte, Novavax and BioNTech all fell Monday after Peter Marks, head of the FDA office that reviews vaccines, resigned on Friday.

Pharmaceutical Technology
MARCH 31, 2025
Non-profit organisations provide an alternative to rescue stalled orphan drugs for biotechs struggling against a lethargic investment climate.

Speaker: Simran Kaur, Co-founder & CEO at Tattva.Health
AI is transforming clinical trials—accelerating drug discovery, optimizing patient recruitment, and improving data analysis. But its impact goes far beyond research. As AI-driven innovation reshapes the clinical trial process, it’s also influencing broader healthcare trends, from personalized medicine to patient outcomes. Join this new webinar featuring Simran Kaur for an insightful discussion on what all of this means for the future of healthcare!

Bio Pharma Dive
MARCH 31, 2025
Accelerate biomarker research with high-quality, expert-curated data to drive precision medicine.
Pharmaceutical Technology
MARCH 31, 2025
Based on the Phase II data, Imbria Pharmaceuticals inerafaxstat could complement existing treatments rather than replace them.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Pharmaceutical Technology
MARCH 31, 2025
The Ukrainian government has instigated rapid cost-cutting measures across the drug retail sector, triggered by reports of overpricing.

Bio Pharma Dive
MARCH 31, 2025
The drug discovery company, which has partnerships with Eli Lilly and Novartis, is developing AI models to design drugs and predict molecular structures.
Pharmaceutical Technology
MARCH 31, 2025
For cell and gene therapy, a consistent supply of quality starting material is vital that is maintained throughout the treatment life cycle.
pharmaphorum
MARCH 31, 2025
The CHMP has advised against EU approval of Eli Lilly's Alzheimer's drug Kisunla, potentially handing an advantage to Eisai/Biogen's rival drug

Speaker: Simran Kaur, Founder & CEO at Tattva Health Inc.
The healthcare landscape is being revolutionized by AI and cutting-edge digital technologies, reshaping how patients receive care and interact with providers. In this webinar led by Simran Kaur, we will explore how AI-driven solutions are enhancing patient communication, improving care quality, and empowering preventive and predictive medicine. You'll also learn how AI is streamlining healthcare processes, helping providers offer more efficient, personalized care and enabling faster, data-driven

Pharmaceutical Technology
MARCH 31, 2025
Despite a haemophilia market bustling with rivals, Qfitlias broad label and favourable dosing regimen could give it an upper hand.

pharmaphorum
MARCH 31, 2025
Novo Nordisk's Ozempic has been shown to improve walking distance in people with diabetes and peripheral artery disease in the STRIDE trial

Pharmaceutical Technology
MARCH 31, 2025
Vertex continues to develop type 1 diabetes therapy, zimislecel, which is being investigated in a Phase III trial.

pharmaphorum
MARCH 31, 2025
Industry organisations are alarmed by the resignation of senior FDA official Peter Marks, citing "misinformation and lies" by HHS Secretary Kennedy

Pharmaceutical Technology
MARCH 31, 2025
Completed biopharma IPOs that raised more than $100 million witnessed an almost twofold increase during 2024.

pharmaphorum
MARCH 31, 2025
Isomorphic Labs has raised a whopping $600 million to refine its artificial intelligence-powered drug discovery engine and advance its pipeline.

Pharmaceutical Commerce
MARCH 31, 2025
Jen Butler, chief commercial officer, Pleio, discusses the importance of balancing technology and personalization in patient support.

pharmaphorum
MARCH 31, 2025
Watch the exclusive interview with Brian Hilberdink, the new head of Boehringer US, as he discusses GLP-1s, DtX, and more in this insightful video discussion.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

Pharmaceutical Commerce
MARCH 31, 2025
Renier Brentjens, MD, PhD outlines the evolution of the space, while offering ways to boost patient access to these treatments.

pharmaphorum
MARCH 31, 2025
Accelerating Breakthroughs in Immunodermatology Drug Development Sara.

Pharmaceutical Commerce
MARCH 31, 2025
The latest news for pharma industry insiders.

pharmaphorum
MARCH 31, 2025
The Home of Accelerating Microbiome R&D Sara.
Pharmaceutical Technology
MARCH 31, 2025
Use of GLP-1RA therapies in CV/CKD disease with T2D will likely rise as physicians view them as addressers of cardiometabolic syndrome.

pharmaphorum
MARCH 31, 2025
At Anthropy 2025: Rebooting Britain, web editor Nicole Raleigh sat down with Dr Sebastian Vaughn, CEO of Phytome Life Sciences, for an en plein air and explorative conversation around the topic of mental health, live onsite at The Eden Project in Cornwall, following the Adelphi sponsored panel, Revolutionary Thinking for Mental Health.

FDA Law Blog
MARCH 31, 2025
By Steven J. Gonzalez On March 31, 2025, U.S. District Judge Sean D. Jordan ordered that FDAs Laboratory Developed Tests (LDT) Final Rule be vacated and set aside, in its entirety. That Rule sought to codify FDAs view that LDTs are medical devices subject to FDA regulation under the Food, Drug, and Cosmetic Act (FDCA) and then phase out, over a four-year period, FDAs purported policy of enforcement discretion for such tests.

pharmaphorum
MARCH 31, 2025
Eli Lilly's lepodisiran has been shown to reduce Lp(a) levels by more than 90% for at least six months with a single injection in a phase 2 trial

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.
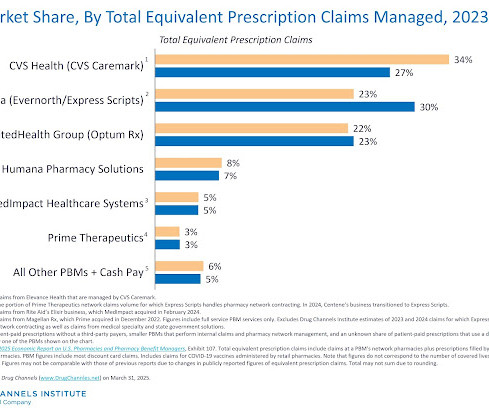
Drug Channels
MARCH 31, 2025
Threes still company in the world of pharmacy benefit managers. For 2024, nearly 80% of all equivalent prescription claims were processed by three familiar companies: the CVS Caremark business of CVS Health, the Express Scripts business of Cigna, and the Optum Rx business of UnitedHealth Group. The names havent changed, but shifting relationships and contract shakeups have altered the plot, with Express Scripts stepping into a new lead role.

XTalks
MARCH 31, 2025
The FDA has granted De Novo authorization to the Visby Medical Womens Sexual Health Test. The PCR-based test detects three of the most common and curable STIs: chlamydia, gonorrhea and trichomoniasis. The single-use test, designed for women and regardless of symptom presence, delivers results in approximately 30 minutes. It includes a self-collected vaginal swab and a powered testing device.

Sciensano
MARCH 31, 2025
Event type: closed meeting Audience: health professionals Description: You are kindly invited to the IQED -Foot Information Meeting of audit 8, organised by Sciensano in collaboration with the Group of Experts IQED -Foot, on behalf of the National Institute of Health and Disability Insurance ( NIHDI ). Programme 18.00 19.30 : Registration and walking dinner 19.30-22.15 : Presentations and discussion 22.30 : End Registration Please register before 10 June using this form Contact If you have any
XTalks
MARCH 31, 2025
Fresenius Kabi received FDA approval for its Prolia and Xgeva biosimilars, Conexxence (denosumab-bnht) and Bomyntra (denosumab-bnht), respectively. The FDA granted authorization to the denosumab biosimilars for all indications corresponding to their reference products. The agency approved Conexxence for use in adult patients at high risk for fractures.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud
Let's personalize your content