Cerevel, in ‘major surprise,’ finds success in late-stage Parkinson’s study
Bio Pharma Dive
APRIL 18, 2024
Analysts had low expectations for Cerevel’s drug tavapadon, which was something of an afterthought in AbbVie’s proposed buyout of the biotech.

Bio Pharma Dive
APRIL 18, 2024
Analysts had low expectations for Cerevel’s drug tavapadon, which was something of an afterthought in AbbVie’s proposed buyout of the biotech.

BioSpace
APRIL 14, 2024
GLP-1 treatments are all the rage in this space, but they aren’t the only approach in development. The pipeline assets highlighted here offer a differentiated approach, potentially increasing efficacy or reducing side effects.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
APRIL 18, 2024
Clearmind Medicine has entered into a licensing agreement with Yissum to develop Generation 3.0 psychedelic compounds for mental disorders.

Rethinking Clinical Trials
APRIL 17, 2024
Dr. Joseph Ross In this Friday’s PCT Grand Rounds, Joseph Ross of Yale University will present “The Yale Open Data Access (YODA) Project: 10 Years of Clinical Trial Data Sharing.” The Grand Rounds session will be held on Friday, April 19, 2024, at 1:00 pm eastern. The YODA Project promotes open science, research transparency, and the sharing of clinical research data to support healthcare research.

Speaker: Simran Kaur, Co-founder & CEO at Tattva Health Inc.
AI is transforming clinical trials—accelerating drug discovery, optimizing patient recruitment, and improving data analysis. But its impact goes far beyond research. As AI-driven innovation reshapes the clinical trial process, it’s also influencing broader healthcare trends, from personalized medicine to patient outcomes. Join this new webinar featuring Simran Kaur for an insightful discussion on what all of this means for the future of healthcare!

Bio Pharma Dive
APRIL 18, 2024
It’s “early innings” in the obesity drug race, said Metsera CEO Clive Meanwell, whose company has acquired a portfolio of medicines it aims to test in combination.

Pharma Times
APRIL 18, 2024
The Tyche model could help clinicians and researchers capture crucial information in images
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.
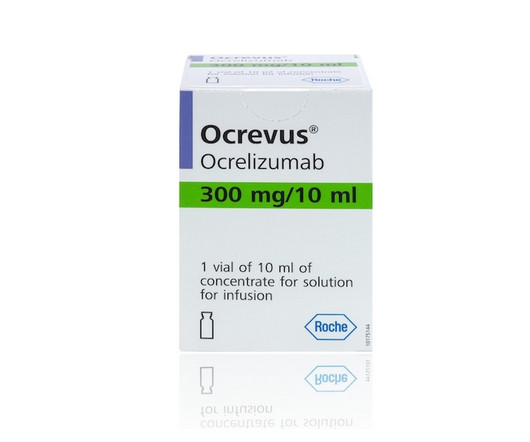
Fierce Pharma
APRIL 17, 2024
One-year data continued to support a more convenient, injectable version of Roche’s blockbuster multiple sclerosis (MS) drug Ocrevus ahead of an FDA decision, the Swiss pharma said. | One-year data continued to support a more convenient, injectable version of Roche’s blockbuster multiple sclerosis drug Ocrevus ahead of an FDA decision, the Swiss pharma said.

Bio Pharma Dive
APRIL 15, 2024
Treatment appeared to result in functional and cognitive gains in people with the neurological disorder. Three participants experienced lower extremity weakness, however.
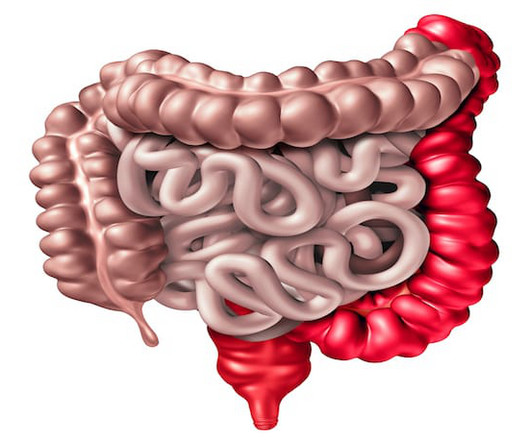
Pharma Times
APRIL 15, 2024
Affecting over 500,000 people in the UK, IBD comprises Crohn’s disease and ulcerative colitis
Pharmaceutical Technology
APRIL 16, 2024
Norgine has sought approval for eflornithine (DFMO) for the treatment of patients with high-risk neuroblastoma (HRNB).

Speaker: Simran Kaur, Founder & CEO at Tattva Health Inc.
The healthcare landscape is being revolutionized by AI and cutting-edge digital technologies, reshaping how patients receive care and interact with providers. In this webinar led by Simran Kaur, we will explore how AI-driven solutions are enhancing patient communication, improving care quality, and empowering preventive and predictive medicine. You'll also learn how AI is streamlining healthcare processes, helping providers offer more efficient, personalized care and enabling faster, data-driven

pharmaphorum
APRIL 16, 2024
A smart sensor developed by Adherium has been cleared by the FDA for use with AstraZeneca's asthma inhaler Airsupra and Breztri for COPD.

Bio Pharma Dive
APRIL 18, 2024
The start of a so-called rolling review formally begins an FDA evaluation of a closely watched drug that’s important to the biotech’s future, but faces questions about its sales potential.

Pharma Times
APRIL 19, 2024
Both will develop cancer vaccine candidates in selected haematological and solid tumour indications
Pharmaceutical Technology
APRIL 16, 2024
The price ceiling policy has been in place for more than 20 years, but it has neither been very successful nor free of consequences for pharma companies.

Pharma Mirror
APRIL 17, 2024
As an authority in radiation oncology, Dr. Khuntia joining the board of directors validates BNCT and the work TAE Life Sciences is doing to make it a mainstay of modern cancer care IRVINE, Calif.- TAE Life Sciences, a pioneer in advancing Boron Neutron Capture Therapy (BNCT) for cancer treatment, proudly announces the appointment of Dr. Deepak “Dee” Khuntia, M.D., FASTRO, to its esteemed Board of Directors.

Bio Pharma Dive
APRIL 17, 2024
The companies plan to launch their copycat version of the blockbuster immune disease drug early next year, per a legal settlement with J&J.

Pharma Times
APRIL 16, 2024
Solid organ transplantation is used to treat end-stage organ failure, including the kidneys
Pharmaceutical Technology
APRIL 18, 2024
Inflammation, cell metabolism and neuronal health are current key areas of focus for dementia researchers, according to Jane Osbourn, chief scientific officer (CSO) at Alchemab.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

Worldwide Clinical Trials
APRIL 15, 2024
As the clinical research landscape continually evolves, the choice of a Contract Research Organization (CRO) partner can significantly impact the success of drug development programs. In fact, recent industry dynamics indicate a considerable shift in preference towards midsize CROs, driven by the need for more personalized, agile, and cost-effective partnerships.

Bio Pharma Dive
APRIL 19, 2024
The new label for Alecensa makes it the first targeted treatment available after surgery for people with ALK-positive lung tumors, who are at high risk for brain metastases.
Pharma Times
APRIL 17, 2024
The findings suggest why children tend to experience milder symptoms compared to older adults

Pharmaceutical Technology
APRIL 17, 2024
LX2006 consists of adeno-associated virus (AAV) vector carrying the frataxin gene, responsible for causing Friedreich’s ataxia.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

Fierce Pharma
APRIL 18, 2024
Novo Nordisk is receiving pushback from a trio of lawmakers over its decision to discontinue its long-acting insulin product Levemir, which was due for a steep discount at the start of the new year | Novo Nordisk is receiving pushback from a trio of lawmakers on its decision to discontinue its long-acting insulin product Levemir, which was due for a steep discount at the start of the new year.

Bio Pharma Dive
APRIL 15, 2024
The results could give Roche’s Columvi an edge over a rival medicine from AbbVie and Genmab, while Regeneron awaits an approval of its own therapy.
Pharma Times
APRIL 16, 2024
Liver cirrhosis, caused by long-term liver damage, is estimated to affect around 100 million people worldwide

Pharmaceutical Technology
APRIL 19, 2024
The US Food and Drug Administration (FDA) has granted fast-track designation to Bayer subsidiary Asklepios BioPharmaceutical’s (AskBio) investigational gene therapy AB-1002, aimed at treating congestive heart failure (CHF).

Speaker: Dr. Ben Locwin - Biopharmaceutical Executive & Healthcare Futurist
What will the future hold for clinical research? A recent draft from the FDA provides valuable insight. In "Optimizing the Dosage of Human Prescription Drugs and Biological Products for the Treatment of Oncologic Diseases," the FDA notes that "targeted therapies demonstrate different dose-response relationships compared to cytotoxic chemotherapy, such that doses below the Maximum Tolerated Dose (MTD) may have similar efficacy to the MTD but with fewer toxicities.

pharmaphorum
APRIL 16, 2024
Researchers from the UK have developed an endoscopic device that uses 3D imaging to look at the stiffness of cells and could diagnose cancer earlier.

Bio Pharma Dive
APRIL 15, 2024
Because live cell and tissue products are extremely sensitive to contamination from microorganisms, viral particles or other airborne impurities, research and manufacturing involving these materials needs to be conducted in a cleanroom environment.

Pharma Times
APRIL 19, 2024
Involving 152 patients, the SENTINEL trial aims to cut lung transplant rejection by up to 50%

Pharmaceutical Technology
APRIL 19, 2024
Paolo Morgese, vice president of European public affairs at the Alliance for Regenerative Medicine, discusses the barriers affecting access to gene therapies.
Let's personalize your content