Verona scores FDA approval for Ohtuvaye as COPD maintenance therapy
Pharmaceutical Technology
JUNE 27, 2024
Ohtuvaye is expected to rake in $1.1bn in global sales from the market for COPD patients with moderate to severe exacerbations by 2029.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
JUNE 27, 2024
Ohtuvaye is expected to rake in $1.1bn in global sales from the market for COPD patients with moderate to severe exacerbations by 2029.
Pharmaceutical Technology
APRIL 28, 2023
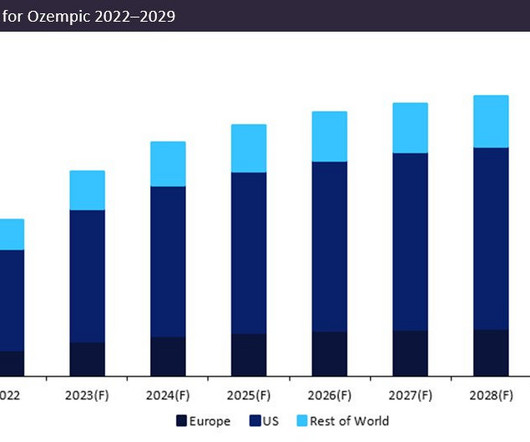
Novo Nordisk’s leading drug Ozempic (semaglutide) is forecast to demonstrate a sales growth of 23% in 2023. Ozempic’s forecast 2023 sales of $12.5bn consolidate its position as the dominant market leader, with projected sales in 2023 54% greater than closest competitor Trulicity (dulaglutide) by Eli Lilly, which anticipates sales of $8bn.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
OCTOBER 23, 2023
The Remicade biosimilar is forecasted to have sales of $2.3bn by 2029 as a subcutaneous form is approved for the US market.
Pharmaceutical Technology
MARCH 30, 2023
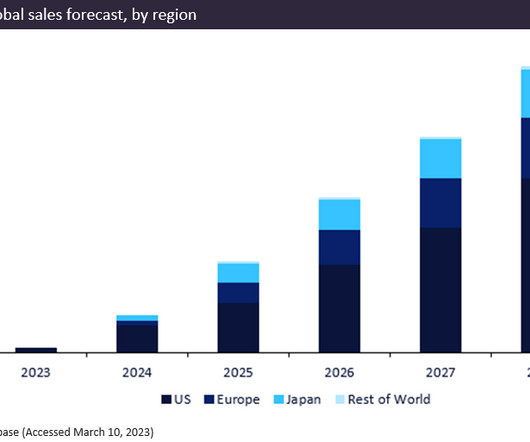
Biogen/Eisai’s newly approved drug, Leqembi (lecanemab), gained FDA approval in January 2023 for the treatment of Alzheimer’s disease. The breakthrough drug is predicted to be a blockbuster, generating total forecast sales of $12.9bn between 2023 and 2028.

XTalks
APRIL 3, 2024
While Soliris had its market launch 17 years ago with $3.2 billion in sales in 2023, Ultomiris was approved in 2018 and generated $3 billion in revenue last year. Voydeya’s FDA approval was based on positive results from the pivotal ALPHA Phase III trial.
Pharmaceutical Technology
MAY 25, 2023
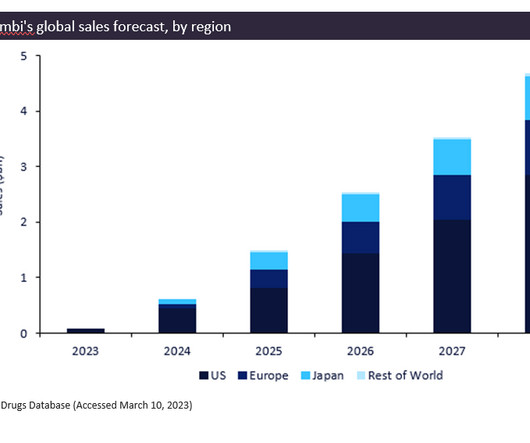
It is already the first proposed high-concentration (100mg/mL), low-volume and citrate-free adalimumab biosimilar in the world to receive marketing authorisation from the European Commission. Yuflyma represents the company’s fifth biosimilar and second anti-TNF biosimilar to receive US FDA approval.
Pharmaceutical Technology
MARCH 30, 2023
Biogen/Eisai’s newly approved drug, Leqembi (lecanemab), gained FDA approval in January 2023 for the treatment of Alzheimer’s disease. The breakthrough drug is predicted to be a blockbuster, generating total forecast sales of $12.9bn between 2023 and 2028.

BioPharma Reporter
APRIL 25, 2024
Back in 2014, AstraZeneca and Merck were the first to receive FDA approval for their PARP inhibitor Lynparza (olaparib) for use in breast cancer gene (BRCA)-mutated metastatic ovarian cancer patients.
XTalks
JUNE 3, 2024
First approved in 2011, Eylea’s market value was estimated to be $8.79 As one of Regeneron’s best-selling drugs, US sales of Eylea were approximately $5.89 First approved in 2022 and winning a subsequent approval in 2023 for macular edema following RVO, Vabysmo exceeded sales expectations earning about $2.7
XTalks
JUNE 21, 2024
While Merck says the vaccine is specifically designed for adults and covers serotypes that cause about 84 percent of IPD in adults 50 years of age and older, it is officially approved for adults 18 years of age and older. Pfizer’s Prevnar 20 was first approved in 2021 for adults 18 years of age and older and has since dominated the market.
XTalks
JUNE 26, 2023
The approval comes after the FDA made several requests for additional data, which lengthened the approval process that began in 2021. Surmodics will manufacture and supply the device and realize revenue from product sales to Abbott as well as a share of profits from Abbott’s third-party sales.
Pharmaceutical Technology
FEBRUARY 8, 2023
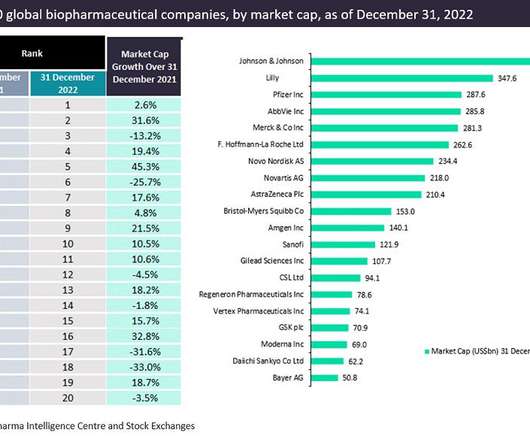
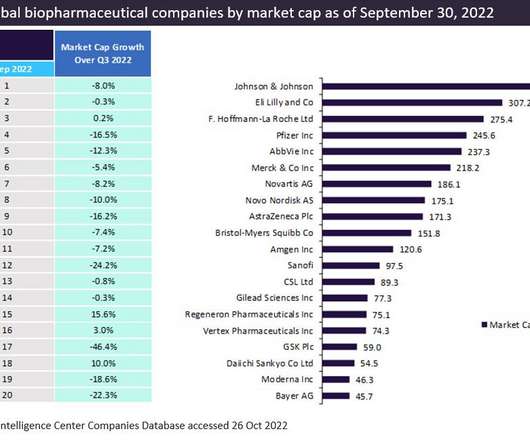
The top 20 companies in the global biopharma industry saw 5.4%* aggregate market capitalization from $3.43 Pfizer maintained its third-place position in the market in 2022, reporting a market capitalization of $287.6 witnessed the biggest market capitalization growth of 45.3% trillion to $3.61 billion on Dec 31, 2022.

XTalks
FEBRUARY 9, 2024
As we delve into the top 15 cardiovascular disease drugs in 2023, guided by comprehensive 2022 sales data, we gain valuable insights into the market trends, drug efficacy and the evolving needs of patients worldwide. Read on to learn more about the top 15 cardiovascular disease drugs in 2023, based on 2022 sales statistics.
XTalks
DECEMBER 19, 2023
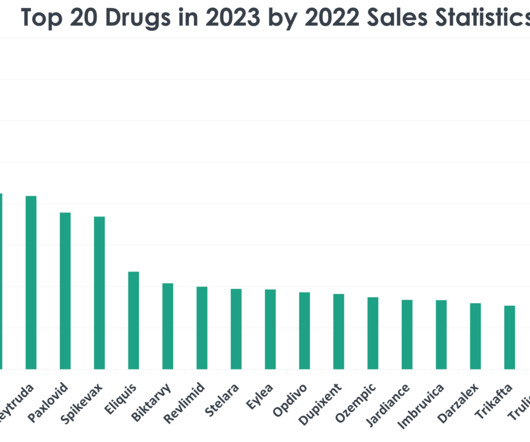
In the fast-evolving pharmaceutical landscape, tracking the top-selling drugs offers valuable insights into trends, healthcare priorities and the dynamics of the global market. As we approach the end of 2023, a retrospective look at the statistics from 2022 reveals the top 20 drugs dominating retail sales. billion in sales in 2022.
Pharmaceutical Technology
APRIL 11, 2023
The US Food and Drug Administration (FDA) has granted final approval to India-based Zydus Lifesciences ‘ 500mg azithromycin tablets for the treatment of bacterial infections. Zydus, which currently has 360 approvals, has filed abbreviated new drug applications for more than 440 drugs since 2003-04.

XTalks
MARCH 28, 2024
Italy-based drugmaker Italfarmaco has won US Food and Drug Administration (FDA) approval for its oral medication Duvyzat (givinostat) for the treatment of Duchenne muscular dystrophy (DMD) in patients six years of age and older. The drug generated domestic sales of $200.4 million last year, of which $131.3
Pharmaceutical Technology
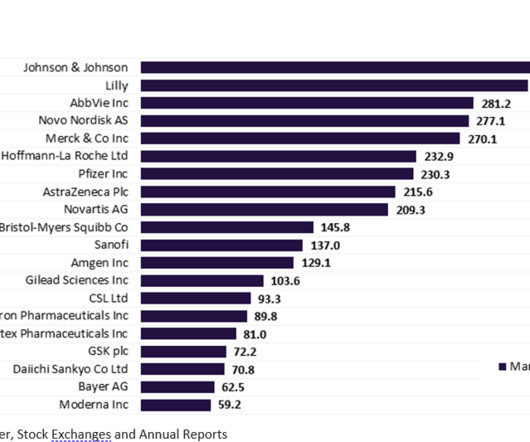
MAY 16, 2023
More than half of the top 20 global biopharmaceutical companies saw a fall in market capitalisation over Q1 2023. decline in total aggregate market capitalisation from $3.61 Bayer reported the highest market capitalisation growth of 23.1% Sanofi and Regeneron’s market capitalisation grew by 12.4% This resulted in a 3.4%
XTalks
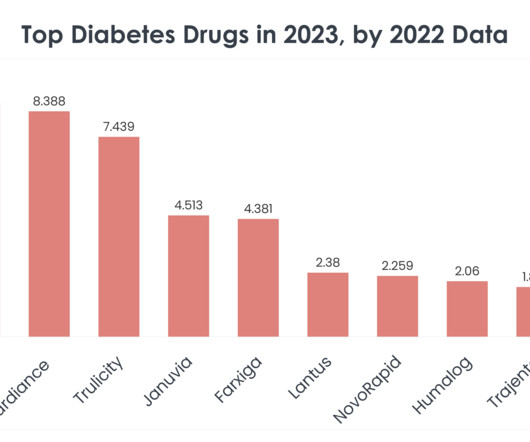
FEBRUARY 8, 2024
Understanding the market dynamics of diabetes treatments becomes crucial for professionals across these industries. The top 15 diabetes drugs in 2023, according to 2022 sales data, reflect the current state of diabetes management and hint at the evolving needs and trends within this critical area of healthcare. billion in Q4 2022.

pharmaphorum
JANUARY 26, 2022
Immunocore has secured a piece of biotech industry history, becoming the first company to get an FDA approval for a cancer therapeutic based on T cell receptor (TCR) technology. The post Immunocore claims first-ever FDA approval for TCR cancer therapy appeared first on.
XTalks
JULY 28, 2021
Merck has scored US Food and Drug Administration (FDA) approval for its next-generation pneumonia vaccine Vaxneuvance that covers 15 different strains of the pneumococcal bacteria that causes the infection. billion in sales in 2020 and has been outselling Merck’s previous shot, Pneumovax 23, by almost five to one.
Pharmaceutical Technology
DECEMBER 20, 2022
The top 20 global biopharmaceutical companies exhibited a downward trend in aggregate market capitalisation by 9.1% This downturn in market cap was attributed to a decline in the demand for Covid-19 vaccines and therapies. Bayer recorded a negative market capitalization growth of 22.3% trillion in Q2 2022 to $3.14

XTalks
JULY 4, 2024
This blog delves into the top 30 drugs to watch in 2024, leveraging insights from their sales performance in 2023. These medications have not only redefined therapeutic standards across various medical conditions but have also showcased impressive sales figures, innovative advancements and expanded indications. billion DKK ($21.1

Pharmaceutical Technology
JUNE 21, 2023
Ketarx will be marketed under the FDA-approved label of ketamine and in dosages that include 10mg/ml, 50mg/ml and 100mg/ml, with the option to boost the concentration and ready-to-administer applications. Ketamine is currently on the FDA’s drug shortage list.

Pharmaceutical Technology
MAY 19, 2023
The US Food and Drug Administration (FDA) has approved AbbVie’s Rinvoq (upadacitinib) for patients with Crohn’s disease who do not respond to TNF blockers, a common immune suppressant treatment for the condition. Data from two Phase III studies, U-EXCEED and U-EXCEL, involving 857 patients was used to support the approval.
Drug Discovery World
FEBRUARY 22, 2024
market capitalisation growth to $3.67 The top 20 global biopharmaceutical companies experienced varied year-on-year (YoY) market capitalisation shifts in 2023 amid the macroeconomic headwinds, steep patent cliffs and the commencement of US drug price negotiations under the Inflation Reduction Act (IRA). billion for 2023.

pharmaphorum
SEPTEMBER 16, 2021
Takeda has had a run of bad luck with its Wave1 pipeline of new drug candidates of late, but can now celebrate a vestry after getting FDA approval for first-in-class lung cancer therapy Exkivity. The post Takeda gets a win for its Wave1 pipeline, as Exkivity nabs FDA approval appeared first on.

Pharmaceutical Technology
OCTOBER 28, 2022
On 24 October, American vaccine developer Vaxcyte shared positive topline data from a Phase I/II study of its multivalent conjugate pneumococcal vaccine VAX-24 , bringing the 24-valent pneumococcal jab one step closer to market. In June 2021, the FDA approved Pfizer’s Prevnar 20 for adult use, although a nod for paediatric use is pending.
Pharmaceutical Technology
MARCH 14, 2023
The agreement is intended for providing commercial support for CTx-1301, after receiving the US Food and Drug Administration (FDA) approval. Additionally, it intends to start a fixed-dose, placebo-controlled, pivotal Phase III trial in ADHD paediatric and adolescent patients in the middle of this year.

XTalks
MARCH 28, 2024
The US Food and Drug Administration (FDA) has given the nod to Winrevair (sotatercept-csrk injection, 45mg, 60mg) for the treatment of adults with pulmonary arterial hypertension (PAH) to increase exercise capacity, improve WHO (World Health Organization) functional class (FC) and reduce the risk of clinical worsening events. billion in 2032.

Pharmaceutical Technology
JANUARY 19, 2023
Acute myeloid leukemia (AML) is part of a market of blood malignancies that commercial cell therapies have not managed to penetrate yet. However, it is possible to target CD33 in the clinic, as evidenced by the FDA approval of Pfizer’s anti-CD33 antibody-drug conjugate Mylotarg (gemtuzumab ozogamicin) in 2000.

XTalks
OCTOBER 4, 2022
Rolvedon’s approval marks Spectrum’s transformation to a commercial-stage company with the opportunity to compete in a $2 billion dollar market, and offers a unique value proposition,” said Tom Riga, President and Chief Executive Officer of Spectrum Pharmaceuticals, in the company’s press release. How Does Rolvedon Work?

XTalks
OCTOBER 25, 2022
Related: AstraZeneca’s Enhertu Gets FDA Approved as First Therapy for HER2-Low Breast Cancer. Imjudo’s approval was based on results from the HIMALAYA Phase III trial , which showed that patients treated with the Imjudo and Imfinzi combination experienced a 22 percent reduction in the risk of death versus sorafenib.
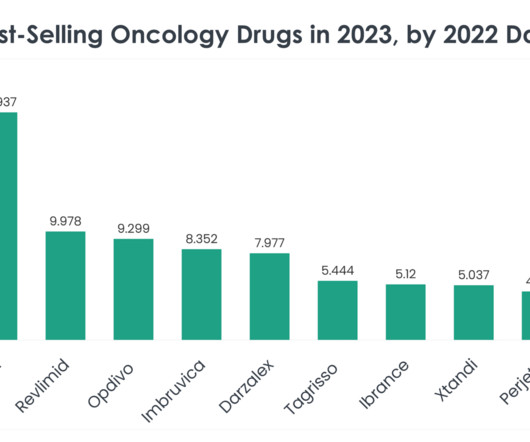
XTalks
FEBRUARY 5, 2024
The top 40 best-selling oncology drugs in 2023, informed by 2022 sales statistics, mark significant milestones in cancer treatment and research. Read on to learn more about the top 40 best-selling oncology drugs in 2023, based on 2022 sales data. Price of Revlimid: A supply of 28 oral 2.5 mg capsules is $24,576.
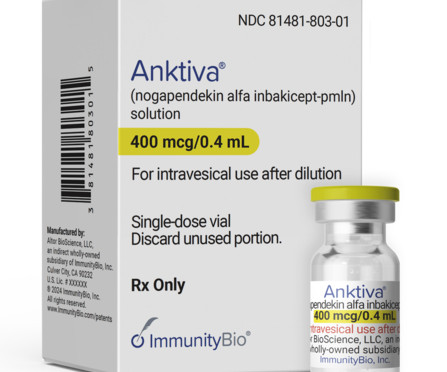
XTalks
APRIL 24, 2024
ImmunityBio’s Anktiva (N-803, or nogapendekin alfa inbakicept-pmln) along with the Bacillus Calmette-Guérin (BCG) vaccine has won US Food and Drug Administration (FDA) approval for the treatment of non-muscle invasive bladder cancer (NMIBC). Anktiva made pharma data intelligence firm Evaluate’s list of top ten drug launches this.

pharmaphorum
AUGUST 4, 2021
Bristol-Myers Squibb’s HDAC inhibitor Istodax has been on the US market for a decade as a treatment for peripheral T-cell lymphoma (PTCL), but will now be withdrawn from sale after a failed phase 3 trial. The post Failed trial nixes another FDA approval, this time for BMS’ Istodax appeared first on.

pharmaphorum
JANUARY 16, 2023
China’s Luye Pharma has claimed its first FDA approval, getting a green light for Rykindo, an injectable formulation of the antipsychotic risperidone given every two weeks. Analysts at GlobalData said last year that they expect LY03010 to launch in 2024 and bring in sales of around $64 million in 2030.

Fierce Pharma
NOVEMBER 2, 2023
Roche’s surge in sales for its long-acting macular degeneration drug Vabysmo showed that patients in the market are open to switching to another treatment. After gaining FDA approval for its high-dose Eylea, Regeneron racked up $43 million in sales in the U.S.

XTalks
FEBRUARY 26, 2024
Xolair comes with a boxed warning for anaphylaxis, which according to the FDA, comes from pre-marketing and post-marketing reports of anaphylaxis that occurred after Xolair administration. In 2018, the FDA granted breakthrough designation for Xolair as a treatment for food allergies. billion in sales last year.
Pharmacy Checkers
NOVEMBER 12, 2020
The new federal rule on drug importation from Canada , drafted pursuant to Section 804 of the FDCA, does not allow for the importation of foreign versions of FDA-approved drugs. It only allows for the importation of FDA-approved drugs. Kesselheim called “ Reputation and Authority FDA and the Fight over U.S.

Pharmaceutical Technology
JUNE 6, 2023
Lotus will purchase global rights for NRX-101 and handle the commercialisation activities of the product in markets outside the US. Lotus and Alvogen will also provide funding for the next registrational trial to support NRX-101’s approval.

Pharmaceutical Technology
JULY 28, 2022
The wAMD and GA markets are anticipated to reach values of $18.8bn and $4.0bn, respectively, in 2031 in the 7MM. The wAMD and GA markets are anticipated to reach values of $18.8bn and $4.0bn, respectively, in 2031 in the 7MM. The therapy received FDA approval in October last year.

pharmaphorum
JUNE 3, 2021
The FDA approval of Alkermes’ new antipsychotic drug Lybalvi couldn’t have gone any better for the company, but it could still face a big challenge positioning the new drug in the highly genericised market. The post After FDA approval, Alkermes plans soft launch for antipsychotic Lybalvi appeared first on.

XTalks
JUNE 6, 2024
billion in 2023, their first non-full year on the market. Despite the more than 60 percent decline in sales, also experienced by Pfizer last year, which Moderna was expecting to be steeper, there were some silver linings. So far in the RSV vaccine arena, GSK’s Arexvy has generated 1.2 billion pounds ($1.5 billion in 2022.
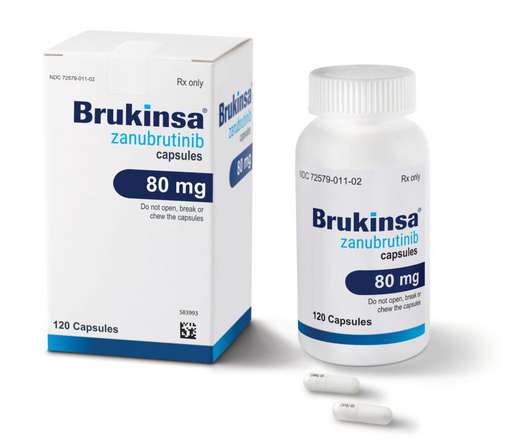
XTalks
JANUARY 26, 2023
This is the fourth US FDA approval that BeiGene has received for Brukinsa since 2019. This includes a greater focus on digital campaigns and diversifying marketing campaigns with race and ethnicity insights.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content