Spark Therapeutics partners with SpliceBio for gene therapy
Pharmaceutical Technology
OCTOBER 17, 2023
SpliceBio will license Spark Therapeutics’ propriety protein splicing platform to develop a gene therapy for renal disease.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

Pharmaceutical Technology
OCTOBER 17, 2023
SpliceBio will license Spark Therapeutics’ propriety protein splicing platform to develop a gene therapy for renal disease.
Pharmaceutical Technology
JUNE 21, 2023
CSL announced the first patient with haemophilia B has been treated with its recently approved gene therapy Hemgenix (etranacogene dezaparvovec). Hemgenix is the first and only gene therapy approved for haemophilia B. per dose, it is the most expensive single-use gene therapy in the US.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
NOVEMBER 9, 2022
On November 2, the Institute for Clinical and Economic Review (ICER) released its updated evidence aimed at measuring the clinical effectiveness and cost of the two haemophilia gene therapies. Known by the brand name Roctavian, BioMarin’s haemophilia A therapy valoctocogene roxaparvovec could be fairly priced in the range of $1.95–1.96

Pharmaceutical Technology
DECEMBER 8, 2022
In December last year, the first centre in Japan received authorisation to administer the therapy, while six regional hospitals are currently approved to offer this treatment. Directed against a cell membrane protein called CD19, Yescarta uses the immune system of the patient to destroy cancer. By Cytiva Thematic.
Pharmaceutical Technology
APRIL 13, 2023
Two gene therapies up for approval this year for sickle cell disease could be cost effective in some cases at a $2 million price point, based on a draft evidence report published by the Institute for Clinical and Economic Review (ICER). Also known as lovo-cel, bluebird bio’s product is a lentiviral gene therapy.

Pharmaceutical Technology
JUNE 15, 2023
Japan-based Astellas Pharma has entered a partnership with Cullgen to discover and develop multiple targeted protein degraders through the latter’s proprietary uSMITE targeted protein degradation platform, in a deal potentially worth $1.9bn. Astellas could pay Cullgen up to $85m upon using its license option regarding the deal.
XTalks
JANUARY 4, 2024
Pfizer has kickstarted the new year with its first-ever gene therapy approval, awarded by Health Canada to the company’s Beqvez (fidanacogene elaparvovec) for the treatment of hemophilia B. Treatment involves replacing the missing FIX clotting protein, traditionally through regular infusions.
pharmaphorum
JANUARY 3, 2023
As December 2022 closed out, Pfizer announced positive top-line results from its phase 3 BENEGENE-2 study evaluating fidanacogene elaparvovec (SPK-9001), its investigational gene therapy for treatment of adult males with moderately severe to severe haemophilia B.

Pharmaceutical Technology
SEPTEMBER 22, 2022
Avrobio has received rare pediatric disease designation from the US Food and Drug Administration (FDA) for its investigational gene therapy, AVR-RD-04, designed to treat cystinosis. AVR-RD-04 works by genetically modify a patient's hematopoietic stem cells (HSCs) to express the gene that encodes the cystinosin protein.

Pharmaceutical Technology
APRIL 26, 2023
MiNA Therapeutics has entered into a research collaboration and option licensing agreement with BioMarin Pharmaceutical to speed up the development of therapeutic ribonucleic acid activation (RNAa) candidates to treat rare genetic diseases. The option licensing agreement is based on early-stage clinical results.

pharmaphorum
NOVEMBER 12, 2020
UCB has made a new foray into the gene therapy space, buying fellow Belgium-based company Handl to get control of its adeno-associated virus (AAV) capsid delivery platform and two research programmes in neurodegenerative diseases. The post UCB revs up its gene therapy drive with Handl acquisition appeared first on.

pharmaphorum
OCTOBER 18, 2022
Eli Lilly has agreed to buy hearing loss gene therapy developer Akouos in a deal that could value the company at around $610 million – if its lead candidate progresses as hoped in the clinic. The buyout – for $12.50 The buyout – for $12.50 There are no approved drug treatments for this type of hearing loss.
Pharmaceutical Technology
AUGUST 23, 2022
It comprises messenger ribonucleic acid (mRNA) that encodes for the initial spike protein of SARS-CoV-2 along with mRNA that encodes for Omicron BA.4/BA.5 5 variant spike protein. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva. On obtaining the FDA EUA, the Omicron BA.4/BA.5-adapted

pharmaphorum
OCTOBER 29, 2020
Already a major player in gene therapy, Novartis has swooped on US startup Vedere Bio in a $280 million deal that builds its position in inherited eye diseases that can lead to blindness. Novartis’ big move in the gene therapy market came when it bought AveXis for $8.7

pharmaphorum
DECEMBER 2, 2021
Novartis has licensed a potential drug for Parkinson’s from Belgium’s UCB that it thinks could be the first oral, disease-modifying drug for the disease – if it can avoid the fate of earlier drugs in the class. The Swiss group is paying $150 million upfront for co-development rights to UCB0599 in a deal that could be worth $1.5

pharmaphorum
JUNE 17, 2021
Tau is a protein that is found in cells of the central nervous system and is involved in the assembly and stabilisation of neuronal microtubules – channels used to transport substances to different parts of the nerve cell. In Alzheimer’s, the protein runs amok, forming tangles that have been linked to cell damage and neuronal death.

pharmaphorum
OCTOBER 3, 2022
DMD is a severe, progressive muscle-wasting genetic condition caused by the lack of a protein called dystrophin. Translarna was the first licensed treatment for DMD which addresses the loss of dystrophin. Translarna was the first licensed treatment for DMD which addresses the loss of dystrophin.
pharmaphorum
JANUARY 13, 2022
Sanofi has joined a growing list of drugmakers going after alpha-synuclein targeting drugs for Parkinson’s disease, licensing a bispecific antibody from South Korea’s ABL Bio in a deal that could be worth more than $1 billion.

The Pharma Data
JUNE 7, 2023
Bayer strengthens gene therapy portfolio with lipid nanoparticle technology from Acuitas Therapeutics Bayer AG is joining forces with Acuitas Therapeutics, Inc., “Accessing state-of-the-art LNP technology through this collaboration will add momentum to our gene editing efforts for the benefit of patients.”

Delveinsight
JANUARY 12, 2021
Medivir, IGM Biosciences enters into an exclusive licensing agreement for Birinapant. Medivir AB has entered into an exclusive licensing agreement with IGM Biosciences to receive global, exclusive development rights for Birinapant. AvantGen Enters into a Licensing Agreement for its Anti-SARS-CoV-2 Antibodies with IGM Biosciences.
pharmaphorum
APRIL 21, 2022
” Alpha-synuclein is a protein that gets misfolded and clumps together in the brains of patients with Parkinson’s and other diseases collectively known as synucleinopathies. Interest in the target has also prompted a flurry of licensing deals.

The Pharma Data
JANUARY 13, 2021
Germany-based Cevec Pharmaceuticals GmbH signed a licensing agreement with Biogen for the use of its proprietary ELEVECTA Technology for the manufacturing of adeno-associated virus (AAV) vectors for gene therapy applications. Few details of the deal were disclosed in the announcement.

The Pharma Data
JANUARY 27, 2021
(Nasdaq: PASG), a genetic medicines company focused on developing transformative therapies for rare, monogenic central nervous system (CNS) disorders, today announced that the U.S. FTD is a debilitating form of early onset dementia that currently has no approved disease-modifying therapies.

Delveinsight
DECEMBER 3, 2020
Roche has got an authorization from the FDA for a more accurate COVID-19 blood test capable of measuring the levels of specific antibodies, which target the cell-unlocking spike protein of coronavirus. Janssen buys a gene therapy asset. Janssen buys a new gene therapy aimed at reversing a severe disease, from Hemera Biosciences.

pharmaphorum
MARCH 31, 2021
Novartis has added to its pipeline of radiopharmaceuticals pipeline once again, licensing a series of agents from iTheranostics that can be used to show where tumours are located in the body. . billion to buy targeted radioligand therapy Lu-PSMA-617 from Endocyte , shortly after encouraging results in a phase 2 prostate cancer trial.

The Pharma Data
FEBRUARY 1, 2021
The FDA issued straight-to-final guidance that provides sponsors of monoclonal antibody and other therapeutic protein COVID-19 treatments with recommendations for potency assays to ensure consistent product quality. The guidance does not cover vaccines, hyperimmune globulins, gene therapies, cell therapies or convalescent plasma.

pharmaphorum
JANUARY 10, 2022
Bayer has bolstered its cell and gene therapy platform by securing access to a CRISPR-based gene-editing platform developed by US biotech Mammoth Biosciences. CRISPR drugs can be used to modify the expression of disease-associated proteins in the body, for example, by correcting a mutation in a specific gene.

Pharmaceutical Technology
AUGUST 26, 2008
. “The inherent advantages of biopharma are that it allows you to pursue therapies against targets that are not small molecule therapies, for example by targeting protein-protein interactions and targeting GPCRs (G-protein coupled receptors),” he says.

The Pharma Data
AUGUST 15, 2021
Food and Drug Administration (FDA) has granted Fast Track designation to VTX-801, Vivet’s clinical-stage gene therapy for the treatment of Wilson Disease – a rare, genetic disorder that reduces the ability of the liver and other tissues to regulate copper levels, causing severe hepatic damage, neurological symptoms, and potentially death.

The Pharma Data
AUGUST 3, 2021
SVP, Chief Medical Officer, Novartis Gene Therapies. “We Additionally, STEER will add to the clinical data and emerging real-world evidence for the use of gene therapy to treat SMA. This route of administration has the potential to open up access for older patients to all the benefits of gene therapy.

XTalks
SEPTEMBER 27, 2022
With a list price of $3 million and a recent accelerated US Food and Drug Administration (FDA) approval, bluebird bio’s gene therapy Skysona (elivaldogene autotemcel, eli-cel), has officially become the world’s most expensive drug. Gene Therapy Zynteglo Wins Landmark FDA Approval for Beta Thalassemia.

pharmaphorum
APRIL 7, 2021
In its second licensing deal in the radio-oncology area in consecutive weeks, Novartis has agreed a $1.3 Novartis has made radioligands one of four pillars of its cancer business, along with cell and gene therapy, targeted drugs and immunotherapeutics, and has been steadily bolting on assets to expand its pipeline in this area.

The Pharma Data
DECEMBER 20, 2020
It is an optimized, non-chemically modified mRNA that codes for the prefusion stabilized full-length spike protein of the SARS-CoV-2 virus. FLT180a is an AAV gene therapy. Sio Gene Therapies reported positive six-month follow-up data from the low-dose cohort of its dose escalation Phase I/II trial of AXO-AAV-GM1.

The Pharma Data
SEPTEMBER 15, 2020
20vPnC showed a safety and tolerability profile that was similar to Prevnar 13 ® Pneumococcal 13-valent Conjugate Vaccine [Diphtheria CRM197 Protein]. Based on the acceptable safety profile and the favorable immune response data, including the 4th dose response data, Pfizer received Breakthrough Therapy Designation.

Pfizer
SEPTEMBER 28, 2022
Monovalent refers to any authorized and approved COVID-19 vaccine that contains or encodes the spike protein of only the Original SARS-CoV-2 virus . Pfizer and Sangamo Therapeutics Announce Phase 3 Trial of Investigational Gene Therapy for Hemophilia A Has Re-Opened Recruitment. COMIRNATY® (COVID-19 Vaccine, mRNA) INDICATION.
The Pharma Data
JUNE 26, 2021
Food and Drug Administration (FDA) has accepted the company’s Biologics License Application (BLA), under Priority Review, for Port Delivery System with ranibizumab (PDS) for the treatment of neovascular or “wet” age-related macular degeneration (nAMD). Roche (SIX: RO, ROG; OTCQX: RHHBY), today announced that the U.S.

XTalks
AUGUST 2, 2023
Johnson & Johnson’s Top 5 Best-Selling Drugs of 2022: 1) Stelara (ustekinumab) Stelara is an immunosuppressant biologic therapy that blocks the IL-12 and IL-23 proteins that play a role in plaque psoriasis and Crohn’s disease. Meanwhile, BioMarin’s gene therapy Roctavian is approved by the FDA for the treatment of hemophilia A.

Pfizer
OCTOBER 19, 2022
Monovalent refers to any authorized and approved COVID-19 vaccine that contains or encodes the spike protein of only the Original SARS-CoV-2 virus. The European Commission will review the CHMP recommendation and is expected to make a final decision soon. Pfizer-BioNTech COVID-19 Vaccine, Bivalent (Original and Omicron BA.4/BA.5) INDICATION.
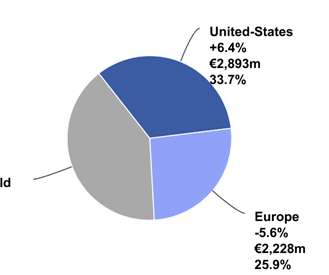
The Pharma Data
APRIL 27, 2021
A new study to select the most appropriate antigen dosage for Phase 3 evaluation of an adjuvanted recombinant protein COVID-19 vaccine candidate (SP0253) was initiated and already completed enrollment. In parallel, development work has commenced against emerging variants, which will be used to inform next stages of the program.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content