Cure Genetics and Frametact seek gene cures for neurological ailments
Pharmaceutical Technology
SEPTEMBER 15, 2023
Cure Genetics has entered a partnership and licensing deal with Frametact to develop gene therapy for familial neurological ailments.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Pharmaceutical Technology
SEPTEMBER 15, 2023
Cure Genetics has entered a partnership and licensing deal with Frametact to develop gene therapy for familial neurological ailments.
Pharmaceutical Technology
OCTOBER 25, 2022
Ast ellas Pharma has announced plans to make a strategic investment to back the development of Taysha Gene Therapies’ adeno-associated virus (AAV) development programmes for Rett syndrome and giant axonal neuropathy (GAN). Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
JUNE 13, 2023
Beacon Therapeutics has kickstarted its entry into the gene therapy field with a $120m Series A financing. Amongst it was AGTC’s lead clinical candidate, AFTC-501, an adeno-associated virus (AAV) gene therapy for XLRP. Beacon Therapeutics is the third gene therapy company Syncona has launched.
Pharmaceutical Technology
JANUARY 5, 2023
Capsida Biotherapeutics and Eli Lilly and Company ’s wholly owned subsidiary Prevail Therapeutics have announced a partnership for the development of non-invasive gene therapies for central nervous system (CNS) diseases. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva. By Cytiva Thematic.
Worldwide Clinical Trials
NOVEMBER 27, 2023
Casgevy, the commercial product formerly known as exa-cel, is administered by taking stem cells out of a patient’s bone marrow and editing a gene in the cells in a laboratory, with the modified cells then infused back into the patient after conditioning treatment to prepare the bone marrow. In June 2023, the U.S.

Pharmaceutical Technology
MARCH 24, 2023
Moderna has entered a strategic partnership with Generation Bio for the development of non-viral genetic medicines. Moderna’s biological and technical expertise will be combined with core technologies of the non-viral genetic medicine platform from Generation Bio.
Pharmaceutical Technology
MAY 24, 2023
It can correct several disease elements associated with PKD, a rare, monogenic red blood cell disorder caused by a mutation in the PKLR gene. It can correct several disease elements associated with PKD, a rare, monogenic red blood cell disorder caused by a mutation in the PKLR gene.

pharmaphorum
OCTOBER 25, 2022
Astellas has been having some safety-related issues with its gene therapy programmes of late, but it shows no signs of lessened enthusiasm for the category – as a new alliance with US biotech Taysha demonstrates. ” The post Astellas makes another gene therapy play, takes stake in Taysha appeared first on.

Pharmaceutical Technology
MARCH 28, 2023
Vertex Pharmaceuticals has signed a new non-exclusive licensing agreement with CRISPR Therapeutics to expedite the development of its hypoimmune cell therapies to treat type 1 diabetes (T1D). The gene-editing technology allows for precise, directed changes to genomic DNA. The system comprises the Cas9 enzyme and a guide RNA.
XTalks
JANUARY 4, 2024
Pfizer has kickstarted the new year with its first-ever gene therapy approval, awarded by Health Canada to the company’s Beqvez (fidanacogene elaparvovec) for the treatment of hemophilia B. There is a significant focus on developing gene therapies as longer-term solutions for the disease.

XTalks
FEBRUARY 4, 2025
Related: FDA Approves New Use for Ozempic to Reduce the Risks of Kidney Disease Maze stands out in the biotech space with its Compass platform, which analyzes genetic data to identify disease-linked variants. For complex diseases like chronic kidney disease (CKD), where genetics play a significant role, such a platform holds promise.

Pharmaceutical Technology
APRIL 26, 2023
MiNA Therapeutics has entered into a research collaboration and option licensing agreement with BioMarin Pharmaceutical to speed up the development of therapeutic ribonucleic acid activation (RNAa) candidates to treat rare genetic diseases. The option licensing agreement is based on early-stage clinical results.

pharmaphorum
FEBRUARY 11, 2022
Data from the first patients enrolled into Regenxbio’s trial of its gene therapy for rare inherited disease mucopolysaccharidosis type I (MPS I) – also known as Hurler syndrome – has shown the first signs of clinical activity. The post Regenxbio expands MPS I gene therapy trial after first look at data appeared first on.

Pharmaceutical Technology
FEBRUARY 9, 2023
ReviR Therapeutics has signed a research collaboration and option-to-license agreement with Asieris Pharmaceuticals to discover new oncology therapeutics. Its initial focus is to develop therapeutics in oncology, CNS, and genetically defined disease indications.
pharmaphorum
JANUARY 3, 2023
As December 2022 closed out, Pfizer announced positive top-line results from its phase 3 BENEGENE-2 study evaluating fidanacogene elaparvovec (SPK-9001), its investigational gene therapy for treatment of adult males with moderately severe to severe haemophilia B. Additional key data is to be presented at scientific conference early this year.

FDA Law Blog
DECEMBER 7, 2022
Valentine — On November 22, 2022, FDA approved CSL Behring’s BLA for Hemgenix (etranacogene dezaparvovec), an AAV-based gene therapy for the treatment of adults with Hemophilia B who currently use Factor IX prophylaxis therapy, have current or historical life-threatening hemorrhage, or have repeated, serious spontaneous bleeding episodes.

Pharmaceutical Technology
FEBRUARY 3, 2023
With scientists fervently developing mRNA vaccines, nucleic acid therapeutics, and viral vector-based gene therapies, clinicians are set to have a growing number of tools available to treat a wide range of conditions, from infectious diseases to genetic disorders and more.

STAT News
MARCH 9, 2023
A decade ago, I had pegged gene and cell therapies as the next frontier. In the spring of 2013, I had just visited a lab focused on gene and cell therapies at Baylor College of Medicine. As an investment banker, I was sure they would someday change how patients were treated. Read the rest…

Pharmaceutical Technology
SEPTEMBER 22, 2022
Avrobio has received rare pediatric disease designation from the US Food and Drug Administration (FDA) for its investigational gene therapy, AVR-RD-04, designed to treat cystinosis. AVR-RD-04 works by genetically modify a patient's hematopoietic stem cells (HSCs) to express the gene that encodes the cystinosin protein.

XTalks
APRIL 24, 2023
Approximately 72 percent of rare diseases are genetic, and around 70 percent of rare genetic diseases emerge in childhood. Eagleton recently spoke on a webinar with his colleagues from Medpace about lessons learned from successful approaches from rare disease and gene therapy product approvals.

FDA Law Blog
MARCH 13, 2023
This meeting will host a panel of rare disease patients, caregivers, and advocates to have a focused discussion on the emerging field of gene therapy (regulated at the FDA by OTP). At least some (if not all) of the panelists will have had experience participating in gene therapy trials. One family’s daughter was the first U.S.
Pharmaceutical Technology
AUGUST 24, 2022
Lacerta Therapeutics has entered a new licensing and research partnership agreement with Eli Lilly and Company subsidiary, Prevail Therapeutics, to discover and develop adeno-associated virus (AAV) capsids for treating central nervous system (CNS) diseases. .

Pharmaceutical Technology
APRIL 6, 2023
Ginkgo Bioworks has strengthened its end-to-end R&D capabilities in gene therapy with the purchase of adeno-associated virus (AAV) capsid discovery and engineering assets from StrideBio, for an undisclosed sum. The new capabilities and IP will be incorporated under Ginkgo’s end-to-end AAV gene therapy development platform.

pharmaphorum
OCTOBER 8, 2020
Scribe Therapeutics, a start-up focusing on gene-editing using CRISPR/Cas9, has burst onto the biotech scene with a $415 million deal with Biogen. It will also use a variation of CRISPR/Cas9 known as CRISPR/Casx which is smaller, making it easier to deliver using viral vectors for example, and more efficient at modifying genes in the body.

pharmaphorum
OCTOBER 26, 2020
Bayer is making clear its ambitions in cell and gene therapy with a $4 billion acquisition of US biotech Asklepios BioPharmaceutical, also known as AskBio. The company’s most advanced programmes are in Pompe disease – a rare genetic disease causing buildup of a sugar molecule inside cells – Parkinson’s disease and congestive heart failure.
XTalks
JUNE 20, 2022
Alnylam’s Onpattro was its first licensed and approved RNAi therapy for ATTR polyneuropathy. Hereditary ATTR has an autosomal dominant pattern of inheritance and is caused by point mutations in the transthyretin ( TTR ) gene that codes for the transthyretin transport protein found in the plasma and cerebrospinal fluid.

pharmaphorum
OCTOBER 18, 2022
Eli Lilly has agreed to buy hearing loss gene therapy developer Akouos in a deal that could value the company at around $610 million – if its lead candidate progresses as hoped in the clinic. The buyout – for $12.50 The buyout – for $12.50 There are no approved drug treatments for this type of hearing loss.

pharmaphorum
APRIL 27, 2021
Dosing can start once again in uniQure’s phase 3 trial of its haemophilia B gene therapy, after the FDA concluded that a case of liver cancer seen in the study was unlikely to be caused by the treatment. . The post FDA lifts hold on uniQure gene therapy after cancer case review appeared first on.

pharmaphorum
JULY 5, 2022
It can be caused by exposure to loud noises – including prolonged use of earbuds at high volumes – as well as cancer treatment, illnesses, genetic mutations, or aging. SNHL is the most common form of hearing loss, account for more than 90% of cases and affecting millions of people worldwide. An estimated 1.57

pharmaphorum
MARCH 24, 2022
GSK has participated in an impressive $175 million Seres C financing for LifeMine led by Fidelity Management & Research Co, paying the company a $70 million license fee along with an undisclosed equity investment as part of a “strategic” partnership focusing initially on three targets.

Delveinsight
JANUARY 12, 2021
Medivir, IGM Biosciences enters into an exclusive licensing agreement for Birinapant. Medivir AB has entered into an exclusive licensing agreement with IGM Biosciences to receive global, exclusive development rights for Birinapant. AvantGen Enters into a Licensing Agreement for its Anti-SARS-CoV-2 Antibodies with IGM Biosciences.

Delveinsight
DECEMBER 8, 2020
KaliVir, Astellas Pharma forms a licensing deal for VET2-L2 oncolytic virus. KaliVir Immunotherapeutics and Astellas Pharma entered into a worldwide exclusive licensing agreement for the development, research, and commercialization of VET2-L2 to widen the horizon of therapeutic approaches available in the Immuno-Oncology market.

pharmaphorum
OCTOBER 3, 2022
DMD is a severe, progressive muscle-wasting genetic condition caused by the lack of a protein called dystrophin. Translarna was the first licensed treatment for DMD which addresses the loss of dystrophin. Translarna was the first licensed treatment for DMD which addresses the loss of dystrophin.
Delveinsight
DECEMBER 17, 2020
Atsena raises USD 55 Million financings for blindness gene therapy. University of Florida (UF) spinout Atsena Therapeutics has completed a USD 55 million financing that will benefit its gene therapy for a common cause of blindness in children into pivotal trials. It is a gene, which codes for a kinase enzyme of the same name.

pharmaphorum
AUGUST 19, 2021
Novartis’ Rydapt has become the first and only licensed treatment for rare and life-threatening blood disorder systemic mastocytosis (SM) to be cleared for routine NHS use, after getting a green light from NICE. It is caused by a genetic mutation in the KIT gene, which triggers the abnormal proliferation of mast cells.

pharmaphorum
OCTOBER 29, 2020
Already a major player in gene therapy, Novartis has swooped on US startup Vedere Bio in a $280 million deal that builds its position in inherited eye diseases that can lead to blindness. Novartis’ big move in the gene therapy market came when it bought AveXis for $8.7 It also gets two preclinical-stage development projects.

XTalks
MARCH 21, 2023
Related: Gene Therapy Clinical Trials for Rare Diseases: 5 Key Takeaways from FDA’s Recent Town Hall Rett syndrome is typically caused by a genetic mutation on the MECP2 gene. Significant development regression associated with the disease occurs after a period of normal development from six to 18 months of age.

XTalks
MAY 1, 2023
Last week, the US Food and Drug Administration (FDA) granted accelerated approval to Biogen’s Qalsody (tofersen) for the treatment of adult patients with amyotrophic lateral sclerosis (ALS) associated with a mutation in the superoxide dismutase 1 ( SOD1 ) gene. This makes Qalsody the first approved treatment to target a genetic cause of ALS.

XTalks
MAY 22, 2023
Related: 5 Food Companies Working With Precision Fermentation Technology How Pairwise Leverages CRISPR Technology CRISPR, or Clustered Regularly Interspaced Short Palindromic Repeats, is a gene-editing technique that can be used to alter the DNA of cells to enhance certain characteristics or reduce less desirable ones.

The Pharma Data
JANUARY 3, 2021
Nasdaq: PASG), a genetic medicines company focused on developing transformative therapies for rare monogenic central nervous system (CNS) disorders, today announced that U.S. GM1, a rare monogenic lysosomal storage disease, is caused by mutations in the GLB1 gene, which encodes the lysosomal enzyme beta -galactosidase (?
XTalks
DECEMBER 13, 2023
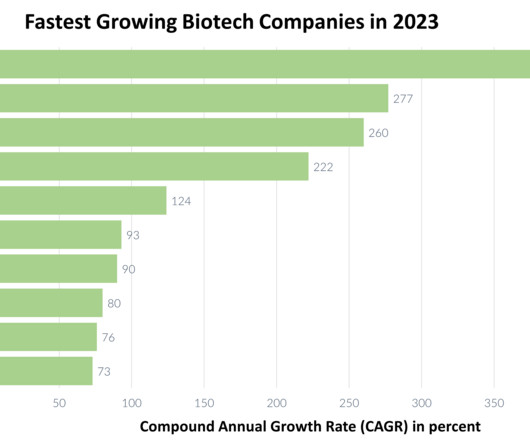
3) Fulgent Genetics Compound annual growth rate: 260 percent Fulgent Genetics, headquartered in California, is a technology-driven company with a strong presence in clinical diagnostics and therapeutic development. The company achieved core revenues of $67 million, reflecting a remarkable 48 perecent year-over-year growth.

pharmaphorum
JANUARY 21, 2021
The world-leading team of academic researchers will use their newly designed technical approach to assess which proteins are selectively produced by the kalirin gene in the human brain, and how this differs from other human tissues. Dr. The significant gap in treatment options leaves many with poor long-term quality of life and disability.
pharmaphorum
MAY 7, 2021
Gene editing firm CRISPR Therapeutics has announced a collaboration with US biotech Nkarta to develop natural killer (NK) cell therapies for cancer. It’s at the forefront of gene editing although the technology has spawned rivals including Intellia Therapeutics, Caribou Biosciences, Sangamo Therapeutics and Mammoth Biosciences.

pharmaphorum
JANUARY 10, 2022
Bayer has bolstered its cell and gene therapy platform by securing access to a CRISPR-based gene-editing platform developed by US biotech Mammoth Biosciences. CRISPR drugs can be used to modify the expression of disease-associated proteins in the body, for example, by correcting a mutation in a specific gene.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content