Digital dilemma in UK’s life sciences job market
Pharmaceutical Technology
SEPTEMBER 8, 2023
In this issue: Digital jobs in UK life sciences, generative AI changes drug discovery, and how robotics is impacting gene therapy manufacturing.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

Pharmaceutical Technology
SEPTEMBER 8, 2023
In this issue: Digital jobs in UK life sciences, generative AI changes drug discovery, and how robotics is impacting gene therapy manufacturing.

Bio Pharma Dive
MAY 6, 2021
Completion of the buyout eases concerns the arrangement might be held up by the FTC, which has signaled it will step up scrutiny of life sciences deals.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
MAY 4, 2023
Forge Biologics and global life sciences company Labcorp have announced a strategic adeno-associated virus (AAV) gene therapy development and manufacturing collaboration. This will improve accessibility to services for AAV-mediated gene therapy programmes.

Pharmaceutical Technology
APRIL 3, 2023
The deal will see Polyplus join the German life science group’s portfolio allowing the latter to leverage expertise in transfection reagents and plasmid DNA for gene therapy. Polyplus, based in Strasbourg, France, produces key components in the production of viral vectors used in cell and gene therapies.

XTalks
MARCH 19, 2025
A 16-year-old patient died after treatment with Elevidys (delandistrogene moxeparvovec), Sarepta Therapeutics gene therapy for Duchenne muscular dystrophy (DMD). Liver injury is a known risk with Elevidys and other gene therapies that use adeno-associated viral (AAV) vectors. Sarepta is reviewing all available data.

Pharmaceutical Technology
MAY 24, 2023
Forge Biologics has joined the public-private collaboration, the Bespoke Gene Therapy Consortium (BGTC), to expedite the development and manufacture of new AAV [adeno-associated virus] gene therapies to treat patients with rare diseases. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.

Pharmaceutical Technology
MARCH 10, 2023
On 10 March, the National Health Service Blood and Transplant (NHSBT) opened a new Clinical Biotechnology Centre (CBC) with the aim of improving the UK’s ability to develop and manufacture cell and gene therapies. The opening of this new facility plays into the UK government’s Life Sciences Industrial Strategy.

Pharmaceutical Technology
MAY 16, 2023
The Foundation for the National Institutes of Health (FNIH) has announced its plans to prioritise eight rare diseases to provide industry standards for manufacturing, preclinical testing and product analytical testing for gene therapy development. This will include pairing up indications with manufacturers amongst the BGTC’s partners.

Bio Pharma Dive
MAY 26, 2022
Bosley, who once led the CRISPR gene editing biotech, will become a venture partner at Advent Life Sciences, which backs a range of early-stage drug companies.

Pharmaceutical Technology
DECEMBER 14, 2022
GSK has entered a strategic partnership with Wave Life Sciences to progress the discovery and development of oligonucleotide therapies for new genetic targets. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva. By Cytiva Thematic.

Pharmaceutical Technology
JUNE 16, 2023
The Abu Dhabi Department of Health (DoH) in the UAE has made a declaration of collaboration with Mass General Brigham’s (MGB) International Center for Genetic Disease (iCGD) to advance life sciences.

Bio Pharma Dive
FEBRUARY 6, 2024
The life sciences investor has backed a number of startups working on technologies for delivering and manufacturing cell and gene medicines.

XTalks
SEPTEMBER 12, 2023
Life science podcasts have emerged as an invaluable tool for building connections with audiences in the digital era. Furthermore, we’ll explore the unique advertising opportunities that this platform offers, positioning brands at the forefront of the life science industry. The result?

XTalks
DECEMBER 20, 2023
As we step into 2024, the life sciences continue to evolve at an unprecedented pace, driven by technological innovation, a deeper understanding of human biology and the application of new technologies in areas like drug development and health wearables.

BioPharma Reporter
FEBRUARY 9, 2023
The Cell and Gene Therapy Catapult (CGT Catapult) has confirmed its involvement in the development of a new Â900m ($1,097m) life science campus in Stevenage, UK: which is set to become one of the largest in Europe.

XTalks
JUNE 14, 2023
Courtney Silverthorn who is an Associate Vice President at the Foundation for the National Institutes of Health (FNIH) and the Director of the Accelerating Medicines Partnership (AMP) program including the Bespoke Gene Therapy Consortium. Subscribe to the Xtalks Life Science Podcast to never miss a new episode.

XTalks
MARCH 25, 2021
Xtalks is proud to announce the launch of the Xtalks Life Science podcast. Subscribe to the Xtalks Life Science Podcast to never miss a new episode. Fresh Conversations About Life Science Topics. She focuses on news relating to the food industry and writes blogs on recruitment and HR in the life sciences.

pharmaphorum
JANUARY 13, 2025
Discover how data science and AI are set to revolutionise the life sciences industry by 2025, with a focus on mRNA technology and CRISPR gene editing. Stay ahead of the curve with these cutting-edge advancements.

XTalks
MARCH 8, 2022
On International Women’s Day, Xtalks is celebrating women’s leadership in the life sciences by highlighting some of the female leaders at the forefront of scientific discovery, as well as the continuing challenges of attaining more equitable representation. Challenges in Women’s Leadership.

Cloudbyz
SEPTEMBER 10, 2023
The challenge of managing and securing sensitive data, including PHI and PII, is significant for life sciences organizations. Additionally, the consequences of data breaches can be severe for life sciences organizations. The challenge is further compounded by the need for fast and accurate analysis of data.
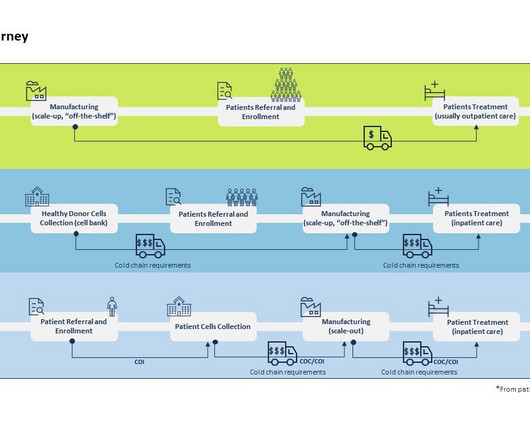
Pharmaceutical Technology
MAY 24, 2023
By Luisa Sterkel & Joana Loureiro , Tenthpin Consultants The promise and potential of cell and gene therapies (CGT) has emerged in the recent past and currently over 1.500 CGT are registered for clinical trials holding great hope for the treatment of challenging and uncurable diseases.

XTalks
APRIL 14, 2021
In this episode of the Xtalks Life Science Podcast, Sarah revisits two Parkinson’s biotech companies — Prevail Therapeutics and Voyager Therapeutics — to see what progress they’ve made in their gene therapy clinical development programs since April 2019. Parkinson’s and Melanoma Share an Amyloid Link, Says New Research.

XTalks
DECEMBER 22, 2021
If the past year is anything to go by, then 2022 will also be a year marked by continued innovations in the life sciences. Here is a look at some life science trends to expect in the coming year, including key themes and topics in the pharmaceutical, biotechnology and medical device industries to keep an eye out for in 2022.

Pharmaceutical Technology
DECEMBER 14, 2022
Merck and Synplogen have signed a non-binding Memorandum of Understanding (MoU) to expedite the development and manufacturing of viral vector-based gene therapy applications. The firms intend to merge their expertise to provide simplified viral vector gene therapy development, production and testing in Japan. By Cytiva Thematic.

XTalks
JANUARY 4, 2023
In this episode, Ayesha talked about some of the trends to look out for in the life sciences in 2023. Read the full article here: 4 Life Sciences Trends for 2023. Subscribe to the Xtalks Life Science Podcast to never miss a new episode.

XTalks
MAY 17, 2023
The Foundation for the National Institutes of Health (FNIH) announced this week that the Accelerating Medicines Partnership Bespoke Gene Therapy Consortium (AMP BGTC) has selected eight rare diseases for its clinical trial portfolio. As such, rare disease patients and their families often face little hope for effective treatments.

pharmaphorum
OCTOBER 22, 2020
Sarepta chief commercial officer Bo Cumbo has left to head up gene therapy venture – AavantiBio – with $107 million in backing from his former employer and three high-profile life sciences investors. Unlike a one-shot gene therapy, omaveloxolone would require continuous dosing to maintain its effects.

XTalks
MAY 22, 2024
In this episode, Ayesha spoke with John Finn, PhD, Chief Scientific Officer at Tome Biosciences , a company developing programmable gene insertion (PGI) technology. PGI is a cutting-edge gene editing technology that allows for the insertion of large sequences of DNA with site-specific precision.

Pharmaceutical Technology
FEBRUARY 13, 2023
Several biotech companies and researchers are now exploring medical devices and gene therapies to address not just common forms of epilepsy, but also rare conditions such as Dravet Syndrome. Meanwhile, others are researching the link between gene variation and different responses to treatments.

XTalks
JANUARY 6, 2025
The CFTR gene itself is complex, with over 2,000 known mutations. The diversity of CF treatments is expanding with investigational therapies such as Spirovants inhaled gene therapy SP-101 in a Phase I/II trial and Kither Biotechs inhaled peptide therapy KIT2014 in a Phase I trial.

XTalks
JANUARY 16, 2025
BIALs Phase II ACTIVATE trial of BIA 28-6156, which targets GBA1 gene mutations, is ongoing, with results expected in 2026 to determine its potential to modify disease progression. With its Phase III trial on the horizon, bemdaneprocel exemplifies the growing potential of cell and gene therapies for Parkinsons disease.
XTalks
SEPTEMBER 9, 2021
The US Food and Drug Administration (FDA) has placed a clinical hold on BioMarin Pharmaceutical’s investigational gene therapy BMN 307 for the rare inherited disease phenylketonuria (PKU) over safety concerns found during preclinical testing. The mice developed the tumors one year after being given BMN 307.
XTalks
JANUARY 4, 2024
Pfizer has kickstarted the new year with its first-ever gene therapy approval, awarded by Health Canada to the company’s Beqvez (fidanacogene elaparvovec) for the treatment of hemophilia B. There is a significant focus on developing gene therapies as longer-term solutions for the disease.

XTalks
JANUARY 24, 2024
In this episode, Ayesha talked about some of the trends in the life sciences to watch out for in 2024. Read the full article here: Life Science Trends to Look Out for in 2024 The weekly podcast is available for streaming every Wednesday on Spotify , Apple Music and wherever you stream your podcasts.

XTalks
JULY 14, 2022
Every gene in a cell is expressed at exact levels due to complex gene regulatory networks. The connections between regulatory genes and their downstream targets have been mapped to some extent — they can be thought of as a subway or metro map, with connections existing between major “hubs.”

pharmaphorum
AUGUST 31, 2022
After a recent approval, there are now three gene therapies available on the US market. In recent years, gene therapy has transitioned from a promising idea to a reality for patients, with many of the severe safety issues that emerged in early iterations of the technology being overcome. from 2021 to 2029.

XTalks
DECEMBER 21, 2022
The past year has been a strong one for life sciences industries, from pivotal gene therapy approvals to continued innovations in the biotech and medical device spaces. Here are some of the top life science trends and topics from 2022. Gene Therapy Approvals. million for a single infusion.

Pharmaceutical Technology
APRIL 21, 2023
IBM will also provide its knowledge to help Moderna explore the potential use cases of quantum technologies in life sciences. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva. Moderna will join the IBM Quantum Network and the IBM Quantum Accelerator programme.
Pharmaceutical Technology
JUNE 4, 2023
Innovation S-curve for the pharmaceutical industry Transcription factors for AAV is a key innovation area in pharmaceutical Adeno-associated virus (AAV) vectors are widely used for gene therapy. There are two main genes in the AAV genome, rep and cap, which encode nine different proteins.

XTalks
JUNE 8, 2021
Novartis’ Zolgensma (onasemnogene abeparvovec) gene therapy has been making significant strides as of late, including dosing of the first Spinal Muscular Atrophy (SMA) patient with the treatment in the UK last week. SMA is caused by mutations in the survival motor neuron 1 ( SMN1) gene, which along with the SMN2 gene, encode the SMN protein.

XTalks
FEBRUARY 2, 2022
The Israeli Society of Gene and Cell Therapy (ISGCT) is hosting its upcoming annual meeting in-person this year to encourage networking and the sharing of information about genetic and cellular therapies. This 2022 meeting will also have a free online panel discussion about gene delivery technologies for therapeutic applications here.
XTalks
DECEMBER 14, 2022
Expanding upon the CRISPR-Cas9 gene editing system, researchers at MIT have designed a new technique called PASTE gene editing that can cut out defective genes and replace them with new genes in a safer and more efficient way. The PASTE gene editing technique was recently published in Nature Biotechnology.

Scienmag
FEBRUARY 5, 2021
Membrane-localized phage proteins may also help revitalize, enhance existing antibiotics A study from the Center for Phage Technology, part of Texas A&M’s College of Agriculture and Life Sciences and Texas A&M AgriLife Research, shows how the “hidden” genes in bacteriophages — types of viruses that infect and destroy (..)

BioPharma Reporter
MARCH 22, 2022
Fujifilm Irvine Scientific, Inc, has announced the acquisition of Shenandoah Biotechnology, a company supplying recombinant proteins to the drug discovery, life science research, and cell and gene therapy markets.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content