mRNA licensing agreements double with million-dollar deals
Pharmaceutical Technology
OCTOBER 7, 2024
mRNA-based vaccines, which instruct cells to produce proteins that can prevent or treat disease, show great promise in precision medicine.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

Pharmaceutical Technology
OCTOBER 7, 2024
mRNA-based vaccines, which instruct cells to produce proteins that can prevent or treat disease, show great promise in precision medicine.
Pharmaceutical Technology
APRIL 25, 2023
German biotechnology firm 3B Pharmaceuticals (3BP) has entered into a licensing agreement with Novartis Innovative Therapies for its fibroblast activation protein (FAP)-targeting peptide technology. 3BP receives an initial payment of $40m, and $425m as development, regulatory and commercial milestone payments.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Pharmaceutical Technology
JUNE 21, 2023
In the past year, Hemgenix has been granted approval by the US Food and Drug Administration (FDA) and has received conditional marketing authorisation from the European Commission (EC) and the UK’s Medicines and Healthcare products Regulatory Agency (MHRA). This followed positive results from CSL’s Phase III HOPE-B trial (NCT03569891).

Pharmaceutical Technology
NOVEMBER 1, 2022
The European Commission (EC) has granted orphan medicinal product designation for Karyopharm Therapeutics and the Menarini Group’s Nexpovio (selinexor) to treat myelofibrosis (MF). An oral inhibitor of exportin 1 (XPO1), Nexpovio works by selectively attaching to hindering the nuclear export protein XPO1.
Pharmaceutical Technology
FEBRUARY 7, 2023
The US Food and Drug Administration (FDA) has accepted Sandoz’s biologics license application (BLA) for its proposed biosimilar denosumab. The BLA covers all indications which are covered by Prolia (denosumab) and Xgeva (denosumab) reference medicines.
The Pharma Data
MARCH 30, 2021
Basel, March 30, 2021 — Novartis has obtained exclusive worldwide rights to develop and commercialize therapeutic applications for a library of Fibroblast Activation Protein (FAP) targeting agents including FAPI-46 and FAPI-74, through an assignment agreement with iTheranostics, Inc., an affiliate of SOFIE Biosciences, Inc.

Pharmaceutical Technology
APRIL 26, 2023
MiNA Therapeutics has entered into a research collaboration and option licensing agreement with BioMarin Pharmaceutical to speed up the development of therapeutic ribonucleic acid activation (RNAa) candidates to treat rare genetic diseases. The option licensing agreement is based on early-stage clinical results.

Pfizer
JULY 26, 2022
Pfizer and BioNTech Advance COVID-19 Vaccine Strategy With Study Start of Next-Generation Vaccine Candidate Based on Enhanced Spike Protein Design. Pfizer and BioNTech Advance COVID-19 Vaccine Strategy With Study Start of Next-Generation Vaccine Candidate Based on Enhanced Spike Protein Design. deliesschef. Tue, 07/26/2022 - 17:41.
Pharmaceutical Technology
AUGUST 23, 2022
It comprises messenger ribonucleic acid (mRNA) that encodes for the initial spike protein of SARS-CoV-2 along with mRNA that encodes for Omicron BA.4/BA.5 5 variant spike protein. The companies have also commenced rolling submission for conditional marketing authorization from the European Medicines Agency (EMA) for this Omicron BA.4/BA.5-adapted

Pharmaceutical Technology
NOVEMBER 9, 2022
CSL Behring has a commercialisation and license agreement to develop EtranaDez. Haemophilia A is caused by insufficient levels of the clotting protein factor VIII, while patients with haemophilia B lack adequate factor IX levels. A fair price for CSL Behring’s haemophilia B treatment etranacogene dezaparvovec would be between $2.93–2.96

pharmaphorum
JANUARY 21, 2021
This is the first project to be funded by the international Psychiatry Consortium, a £4 million collaboration between seven global pharmaceutical companies, and two leading research charities, convened and managed by the Medicines Discovery Catapult, that supports high-value drug discovery projects in this area of unmet patient need.

pharmaphorum
JULY 8, 2022
Swedish rare disease specialist Sobi is paying $55 million upfront to license rights to ADC Therapeutics’ lymphoma therapy Zynlonta – approved in the US last year – in Europe and other international markets. The post Sobi bolsters blood division via $435m ADC licensing deal appeared first on.

pharmaphorum
JANUARY 11, 2023
Anima uses its mRNA Lightning platform to discover small molecules and determine their mechanisms of action for diseases previously deemed undruggable, with scientists searching for compounds that target proteins with roles in regulating mRNA, so as to affect disease states through post-transcriptional regulation (i.e.

The Pharma Data
JANUARY 6, 2021
CAMBRIDGE, England & WETHERBY, England–( BUSINESS WIRE )– Avacta Group plc (AIM: AVCT), the developer of innovative cancer therapies and diagnostics based on its proprietary Affimer ® and pre|CISION platforms, is pleased to announce that it has entered into a license agreement with POINT Biopharma Inc.
XTalks
JANUARY 29, 2021
They are both checkpoint inhibitors targeted towards programmed cell death protein 1 (PD-1 inhibitors), a protein that is responsible for downregulating the immune system and preventing it from killing cancer cells. Biosimilars are increasingly becoming the standard of care and both have several licensed biosimilar drugs (e.g.

The Pharma Data
OCTOBER 27, 2021
NYSE and TASE: TEVA) and MODAG GmbH today announced a strategic collaboration on the exclusive worldwide licensing and development of MODAG’s lead compound anle138b and a related compound, sery433. synuclein protein, mainly in oligodendroglial cells (glial cytoplasmic inclusions) and also in certain nerve cells.

Pfizer
SEPTEMBER 28, 2022
Pfizer and BioNTech Complete Submission to European Medicines Agency for Omicron BA.4/BA.5-Adapted Pfizer and BioNTech Complete Submission to European Medicines Agency for Omicron BA.4/BA.5-Adapted 5-Adapted Bivalent Vaccine Booster in Children 5 Through 11 Years of Age. Tue, 09/27/2022 - 16:15. Wednesday, September 28, 2022 - 11:45am.

pharmaphorum
DECEMBER 2, 2021
GlaxoSmithKline has formed a five-year partnership with Oxford University to set up a new institute that will apply machine learning and functional genomics to the discovery of new medicines. Most notable is GSK’s $700 million upfront deal in the summer to license two drugs for neurological disease developed by US biotech Alector.

Drug Discovery Today
APRIL 22, 2020
Cambridge, UK, and Brisbane, CA, 21 April 2020 Mogrify Ltd (Mogrify®), a UK company aiming to transform the development of cell therapies by the systematic discovery of novel cell conversions, and Sangamo Therapeutics (Sangamo) (Nasdaq: SGMO), a genomic medicine company, today announced that they have executed a collaboration and exclusive license (..)

pharmaphorum
DECEMBER 21, 2021
The US biotech’s other main platform is XTEN, which adds side chains to existing molecules to extend their activity in the body and prevent off-target effects, and has already been licensed to other drugmakers including Biogen and Merck & Co. Sanofi’s other acquisitions this year were the $3.2 billion upfront.

pharmaphorum
DECEMBER 21, 2020
Compared with many of the established names in pharma these companies are young upstarts but they have managed to achieve what other big names in the industry have failed to do and harness the power of mRNA to make medicine. In this case that is the “spike” protein seen on the surface of the coronavirus that it uses to invade host cells.

XTalks
DECEMBER 20, 2021
It has also applied for regulatory licensing in the UK, European Union (EU), Australia, Singapore, India, United Arab Emirates (UAE), New Zealand and Japan. Covovax is a recombinant protein-based vaccine and can be regarded as a more “traditional” vaccine as it isn’t genetic-based like mRNA or viral vector vaccines. Gavi secured 1.1
pharmaphorum
MARCH 8, 2021
The EMA’s human medicines committee has said two Eli Lilly antibodies are effective in people with COVID-19 who are at risk of developing severe disease, clearing the way for use in EU member states.
XTalks
JANUARY 4, 2024
Hemophilia B, also known as congenital Factor IX (FIX) deficiency or Christmas disease, is a rare genetic bleeding disorder caused by a deficiency or defect in the FIX clotting protein. Treatment involves replacing the missing FIX clotting protein, traditionally through regular infusions. Priced at $3.5

pharmaphorum
FEBRUARY 4, 2022
” Nuvaxovid was approved by the Medicines and Healthcare products Regulatory Agency (MHRA) yesterday, becoming the fifth COVID-19 vaccine to be authorised in the UK. In August 2020, the UK government agreed a deal with Novavax to purchase 60 million doses of the vaccine.

pharmaphorum
JULY 5, 2022
Cell therapy specialist Mogrify has struck a deal with Japanese drugmaker Astellas to look at ways to deploy regenerative medicine to treat hearing loss caused by factors such as chronic exposure to loud noises. An estimated 1.57

pharmaphorum
JUNE 4, 2021
The Noria/PSMA deal gives the drugmaker a preclinical-stage alpha radionuclide compound based on actinium-225 and a small molecule directed towards prostate-specific membrane antigen (PSMA), a transmembrane protein overexpressed in prostate cancer.

Pharmaceutical Technology
AUGUST 26, 2008
As understanding of biological systems has forged ahead, pharmaceutical companies have made increasing use of biotechnology in discovering and manufacturing new medicines. ” Today most big pharmaceutical companies are pursuing biotechnology either in house or by using an in-licensing strategy.

The Pharma Data
SEPTEMBER 8, 2020
Fourteen newly licensed medicines have been accepted for use on NHS Scotland by the Scottish Medicines Consortium (SMC), bringing a raft of new treatment options for conditions including epilepsy, depression and cancer.

pharmaphorum
OCTOBER 3, 2022
DMD is a severe, progressive muscle-wasting genetic condition caused by the lack of a protein called dystrophin. Translarna was the first licensed treatment for DMD which addresses the loss of dystrophin. Eventually, they will need assistance with breathing.
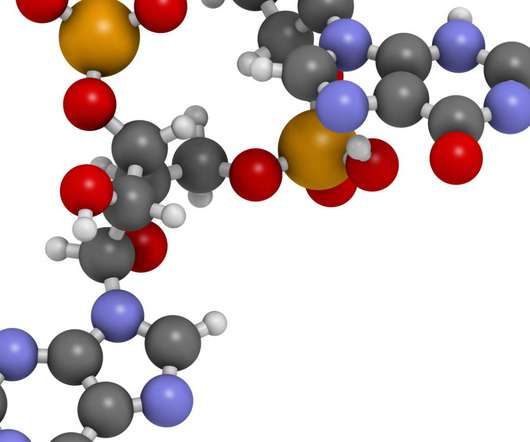
Pharmaceutical Technology
FEBRUARY 14, 2023
Alkaloids, natural dyes, drugs, proteins, and enzymes are some of the important classes of natural heterocyclic compounds essential for human life. The most common heteroatoms are nitrogen (N), oxygen (O) and sulphur (S). Its primary focus is on cancer, cardiovascular, immunology and fibrotic therapeutic projects.
pharmaphorum
JANUARY 3, 2023
SPK-9001 is a novel, investigation vector containing a bio-engineered adeno-associated virus (AAV) capsid (protein shell) and a high-activity human coagulation FIX gene. Additional key data is to be presented at scientific conference early this year.

pharmaphorum
SEPTEMBER 13, 2022
.” If Lamzede is approved by the FDA, it will be another milestone for Chiesi’s two-year-old rare disease division , based in Boston, which was formed to diversify away from the company’s traditional focus on respiratory medicines and further its ambition to grow in the US.

The Pharma Data
AUGUST 17, 2020
. (“Dragonfly”), today announced that they have entered into a definitive agreement under which Bristol Myers Squibb will be granted the global exclusive license to Dragonfly’s interleukin-12 (IL-12) investigational immunotherapy program, including its extended half-life cytokine DF6002. About Bristol Myers Squibb.

The Pharma Data
JUNE 14, 2023
Cedilla’s small molecules conditionally modulate the protein complex in its functional state resulting in highly selective inhibition. Cedilla will also be eligible to receive royalties on medicines based on their technology commercialized by Bayer. Financial details have not been disclosed.

The Pharma Data
NOVEMBER 29, 2020
Binding to the MDM2 protein with high affinity, MDM2 inhibitor blocks the MDM2-p53 interactions and restore the tumor-suppressing activity of p53. Binding to the MDM2 protein with high affinity, MDM2 inhibitor blocks the MDM2-p53 interactions and restore the tumor-suppressing activity of p53. SUZHOU, China and ROCKVILLE, Md. ,

The Pharma Data
DECEMBER 13, 2020
Relay Therapeutics (Nasdaq: RLAY) is a clinical-stage precision medicines company transforming the drug discovery process with the goal of bringing life-changing therapies to patients. . CAMBRIDGE, Mass., 14, 2020 (GLOBE NEWSWIRE) — Relay Therapeutics, Inc. Cautionary Note Regarding Forward-Looking Statements.

The Pharma Data
JANUARY 24, 2021
“We are delighted to be collaborating with the world-renowned researchers at BIDMC, as their cutting-edge research and development work is crucial in bringing potentially life-saving medicines to patients in need of new treatment options.” ZUG, Switzerland , Jan. Chief Medical Officer of Quercis Pharma.

pharmaphorum
JULY 18, 2022
“It is programmed and coded for the production of the full-length spike protein of the coronavirus. It then elicits an immune response against that spike protein. We’ve been inspected by the European Medicines Agency (EMA) and passed the Good Manufacturing Practice (GMP) inspection.
XTalks
JULY 17, 2023
In a significant development, Astellas Pharma announced that the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA) had accepted their applications for Priority Review and Marketing Authorization respectively for zolbetuximab. This investigational first-in-class Claudin 18.2 How Does Zolbetuximab Work?
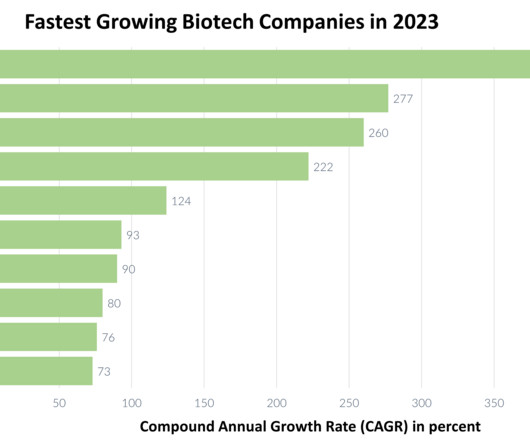
XTalks
DECEMBER 13, 2023
These efforts encompass programs related to the shingles vaccine, Tdap vaccine and plague vaccine, reinforcing Dynavax’s commitment to advancing the field of vaccines and medicine. Alpine Immune Sciences has recently announced an augmentation in its R&D investment. million and $38.8 million, respectively, as compared to $17.6

XTalks
APRIL 3, 2024
The RBCs in circulation are marked by proteins in the complement system for removal by the spleen and liver. Through its Alexion deal, AstraZeneca is looking to expand into other rare diseases, including various forms of amyloidosis for which it has secured licensing deals and buyouts.

The Pharma Data
JANUARY 5, 2021
Targeting RNA is believed to be a way to develop therapeutics for so-called undruggable proteins. This is because many proteins do not have small-molecule binding sites. So therapeutics that focus on RNA are designed to stop the DNA from coding for disease-causing proteins.

XTalks
JANUARY 28, 2022
Immunocore received Breakthrough Device Designation for Kimmtrak from the FDA in February 2021 followed by acceptance of its Biologics License Application (BLA) several months later by both US and European regulators. Kimmtrak is a bispecific protein that contains a soluble T cell receptor fused to an anti-CD3 immune-effector function.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content