Pharma companies must adapt to keep pace with AI developments, say experts
Pharmaceutical Technology
OCTOBER 25, 2024
At the Outsourcing in Clinical Trials Conference, key opinion leaders shared predictions for workflow changes due to AI.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

Pharmaceutical Technology
OCTOBER 25, 2024
At the Outsourcing in Clinical Trials Conference, key opinion leaders shared predictions for workflow changes due to AI.

Bio Pharma Dive
SEPTEMBER 2, 2021
The pharma company is in a tight race with rivals, including GSK, to become the first with an approved shot for the infectious disease.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

AuroBlog - Aurous Healthcare Clinical Trials blog
AUGUST 3, 2023
The Institute is now looking for partners in pharma companies to pursue further clinical development of the findings. Researchers at the Indian Institute of Science (IISc) have found that picolinic acid, a natural compound produced by mammalian cells, can block several disease-causing viruses such as SARS-CoV-2 and influenza A viruses.

Bio Pharma Dive
OCTOBER 1, 2024
Medical meetings often feature important clinical trial results, making them barometers of biotech and pharma companies' research progress. Here’s a list of conferences to watch in 2025.

AuroBlog - Aurous Healthcare Clinical Trials blog
MARCH 29, 2023
Indian healthcare providers and pharma companies pace up their efforts with early detection and research for new medicines to combat the spread of tuberculosis as drug resistance concerns emerge. TB is a significant public health threat, with an estimated 10 million annual cases. India shares the highest TB burden with 2.69

Pharma Times
MARCH 24, 2021
Report outlines two cases for using synthetic data in the clinical trial process
Pharmaceutical Technology
MAY 24, 2023
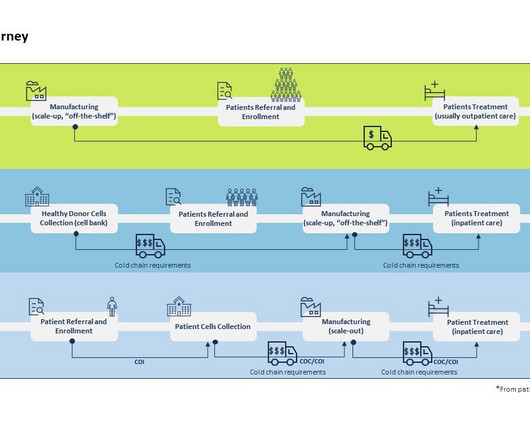
By Luisa Sterkel & Joana Loureiro , Tenthpin Consultants The promise and potential of cell and gene therapies (CGT) has emerged in the recent past and currently over 1.500 CGT are registered for clinical trials holding great hope for the treatment of challenging and uncurable diseases.
Outsourcing Pharma
OCTOBER 15, 2024
The big pharma company Sanofi has discontinued a phase 2 trial of U.S. partner Denali Therapeuticsâ oditrasertib for the treatment of multiple sclerosis after the drug failed to perform.

Bio Pharma Dive
OCTOBER 28, 2022
Disappointing study results have led the French pharma company to take a $1.6 billion impairment charge and plan new early-stage trials for a drug executives once called a potential “foundation” for newer immunotherapy combinations.

Pharmaceutical Technology
JANUARY 16, 2023
Moreover, Insilico Medicine’s INS018_055, an AI-designed candidate, has produced positive topline results in its Phase I clinical trial for idiopathic pulmonary fibrosis (IPF) treatment. In addition to collaborating with AI-technology providers, pharma companies are also engaging in vertical integration.

World of DTC Marketing
JANUARY 11, 2022
Further, 62% of HCPs said that the most significant area where pharma representatives can add value is, by understanding the needs of HCPs and sharing only relevant content with them to make the interactions more insightful. At the same time, marketing emails are among the top 5 channels used by pharma companies to engage HCPs.
Pharmaceutical Technology
MAY 24, 2024
Shares in the pharma company rose 40% in premarket trading.

Pharmaceutical Technology
MARCH 6, 2023
Incannex Healthcare has collaborated with New Jersey-based pharma company Catalent for the development and manufacturing of a cGMP-grade psilocybin drug product for clinical trials and potential commercial use. The latest development follows an internal analysis of the preliminary results of the Phase II PsiGAD clinical trial.
Pharmaceutical Technology
FEBRUARY 2, 2023
Artificial intelligence (AI) continues to be in the news, and as pharma companies invest more to make sure they’re ahead of the game, international agencies have also started to catch up with regulations for AI-led healthcare research. We also explore how AI is being used to design digital twins for clinical trials.
pharmaphorum
MARCH 28, 2022
Built under a government tender, AION Labs comprises four large international pharma companies – AstraZeneca , Merck, Pfizer , and Teva – and another core partner, the Israel Biotech Fund. Yerushalmi states there are numerous ways AI can support the pharma process – from clinical trials to patient stratification.
pharmaphorum
MARCH 12, 2021
COVID-19 has transformed the way we think about patient participation in trial design – but there are still many barriers to overcome in improving engagement. Speakers at the NIHR’s recent virtual event for the pharma industry discussed best practices for bringing patient insights into research.

XTalks
DECEMBER 4, 2024
Jupiter Neurosciences, a clinical-stage pharma company specializing in neuroinflammation, made its public market debut with an initial public offering (IPO) on the Nasdaq Capital Market under the symbol “JUNS.” ” The company priced 2,750,000 shares of common stock at $4.00

AuroBlog - Aurous Healthcare Clinical Trials blog
APRIL 8, 2024
The Drugs Technical Advisory Board (DTAB) has suggested to the ministry of health and family welfare (MoHFW) to write to the ministry of corporate affairs to mandate the pharma companies to spend almost one per cent of their net profit for providing free medicines in medicine banks as part of the Corporate Social Responsibility (CSR). […]

AuroBlog - Aurous Healthcare Clinical Trials blog
MARCH 17, 2024
The Department of Pharmaceuticals (DoP) has issued a new Uniform Code for Pharmaceutical Marketing Practices (UCPMP) 2024, permitting pharma companies to provide brand reminders such as informational and education items and free samples to medical professionals with restrictions on sample packs and total value.

Intouch Solutions
OCTOBER 13, 2023
Pharmaceutical companies can sponsor as well as provide these leaders. Pharmaceutical companies can increase diversity in their clinical trials. Companies can also support pre-existing SDOH research initiatives. Studies should include as detailed SDOH data as possible.

AuroBlog - Aurous Healthcare Clinical Trials blog
APRIL 11, 2023
The pharma companies’ thrust on R&D to develop better medicines and the demand in hospitals for advanced medical technologies driven by artificial intelligence, remote patient monitoring and wearables are transforming patient care.

AuroBlog - Aurous Healthcare Clinical Trials blog
JANUARY 12, 2023
Indian pharma sees immense value in US FDA’s insistence for aluminum content and labelling recommendations for the Small Volume Parenterals Drug Products and Pharmacy Bulk Packages. Pharma companies in the country have increasingly made a shift to nutritional based prescription and over the counter products.

Pharmaceutical Technology
JUNE 15, 2023
But before pharmaceutical companies can go to market with a breakthrough drug, they need to ensure safety and efficacy through clinical trials. Pharma R&D teams are solving this problem by leveraging the power of artificial intelligence (AI) in clinical trials to save time and money.

AuroBlog - Aurous Healthcare Clinical Trials blog
JULY 11, 2023
The Delhi High Court has granted ten days’ time to the Government of India and the nation’s drug regulator to file a counter affidavit on the petitions filed by almost 28 pharma companies against the order prohibiting manufacturing, distribution and sale of 14 FDCs licensed prior to the year 1988, in the beginning of June. […]

World of DTC Marketing
NOVEMBER 27, 2020
Pfizer and Moderna have provided data from their large-scale Phase 3 trials only via news releases , which contained the highly promising news that both vaccines were 90 percent effective or more and have not presented any serious safety concerns. The results of trials were announced in press releases rather than peer-reviewed papers.

AuroBlog - Aurous Healthcare Clinical Trials blog
JULY 16, 2023
At least seven more pharma companies and a federation of pharma manufacturers have approached the Delhi High Court against the Central Government’s order in the beginning of June prohibiting manufacturing, distribution and sale of 14 fixed dose combinations (FDCs) licensed prior to the year 1988.

pharmaphorum
JANUARY 17, 2022
The adoption of decentralised activities was a major factor in allowing clinical trials to continue during the early stages of the pandemic. Ben Hargreaves examines whether this adoption looks set to become a permanent feature of clinical trials moving into the future, rather than just supplying the tools to a temporary need.

World of DTC Marketing
FEBRUARY 7, 2022
At 99% of pharma companies, that's a huge no-no. On top of this, “the company also revealed that the Federal Trade Commission and the Securities and Exchange Commission had opened two new, separate investigations into the troubled biotech. At 99% of pharma companies, that’s a huge no-no.

pharmaphorum
FEBRUARY 7, 2022
The last three years in pharma have seen a growing awareness of and dissatisfaction with a trend that has plagued the industry for its whole life: Nondiverse and unrepresentative clinical trials, which lead to incomplete data and compounded health disparity for minority populations.

pharmaphorum
SEPTEMBER 2, 2020
But many patients still do not respond to therapies such as Merck & Co’s anti PD-1 therapy Keytruda (pembrolizumab), or go on to develop progressive disease, and pharma companies are looking for new second-line therapies. A total of 40 patients are planned to be enrolled.

pharmaphorum
FEBRUARY 11, 2021
Pharma companies face many challenges when involving patients in the design of clinical trials – but doing so can have huge benefits further down the line, improving the sustainability and quality of research. During the course of my treatments and my trials, and afterwards, I was the expert in how I wanted to manage my life.”.

pharmaphorum
OCTOBER 16, 2020
The trial – run by Boehringer Ingelheim and Yale University – will test a smart bathroom scale device that has cardiac monitoring built in, as well as an app to help patients improve their diet and lose weight and a digital assistant designed to motivate them to actively manage their health. “The

pharmaphorum
AUGUST 24, 2022
Diversity in clinical trials is a “scientific imperative”, but how can industry bridge the gap between “why” and “how”? Removing barriers to clinical trial participation for underserved groups is an essential part of addressing health inequalities. Clinical trial participation, she went on, was vital. Why, not how.

AuroBlog - Aurous Healthcare Clinical Trials blog
FEBRUARY 11, 2025
Yet India needs to develop strategies to mitigate the impact of USAID funding freeze for Indian pharma companies. This is even as a large section of companies exporting to the US noted that even if foreign assistance is […]

Pharmaceutical Technology
APRIL 13, 2023
Takeda’s announcement underlines the risk associated with gene therapy R&D at the preclinical stage and the fact that many current AAV programs are unlikely to reach late-stage trials. Takeda’s pivot away from AAV gene therapy follows a similar action taken by Pfizer in January 2023 to externalise its gene therapy R&D.

pharmaphorum
JUNE 14, 2022
Despite more treatment options being a positive, Ben Hargreaves finds that this has raised issues over single-region clinical trials, leading to the FDA rejecting certain treatments and clarifying what is required for approval. Only last month , the US company Merck agreed to pay a potential $1.4 Single-region concerns.
pharmaphorum
JANUARY 24, 2022
The UK scientists that ran one of the largest trials of experimental COVID-19 drugs have formed a non-profit company that will apply the methodology to other disease areas – with $6.8 ” Those trials also need to be large, involve a diverse range of individuals, and able to be run at reduced cost, said Landray. .
pharmaphorum
NOVEMBER 11, 2022
The lack of diversity in clinical trials has been a topic of debate for decades, but was thrust into the spotlight as the impact of the pandemic on poorer, less educated and ethnically diverse populations became even more apparent. Zeroing in on trials with only US sites is a little more encouraging, but the trends are in the same direction.

World of DTC Marketing
AUGUST 9, 2021
HCPs don’t necessarily want pharma trying to sell products via social media. The best approach is to use social media to connect HCPs to clincial information and drug trials. A leading pharma company recently sponsored a series of Q&A around the use of their new cancer drug.
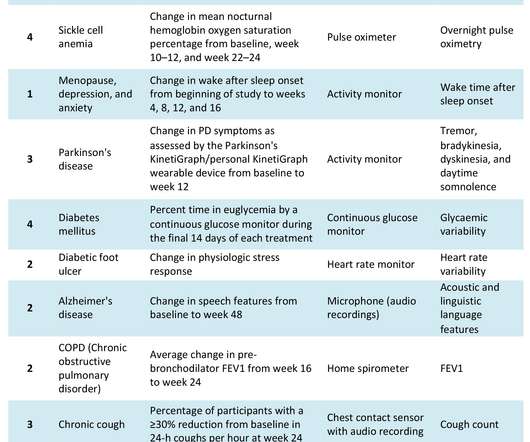
pharmaphorum
JULY 8, 2022
The COVID-19 pandemic has catalysed significant changes in the way pharma develops drugs, particularly in the clinical trial space. Hybrid or decentralised clinical trials (DCTs) have gained traction as technology, infrastructure and knowledge have evolved to support their use. Source: Izmailova et al, 2017.

pharmaphorum
JUNE 11, 2021
A digital therapeutic aimed at people with chronic lower back pain based on virtual reality (VR) has shown encouraging results in a clinical trial, helping patients manage the fear of movement that can reduce their activity and impede recovery. The post Trial finds Orion digital therapeutic helps chronic pain sufferers appeared first on.

pharmaphorum
FEBRUARY 1, 2022
Across the industry, pharma companies are turning to AI and real-world data to address many of the challenges of running clinical trials. Can the combined potential of new AI technologies and real-world patient data hold the key to overcoming the challenges in clinical trial design that have historically led to trial failure?
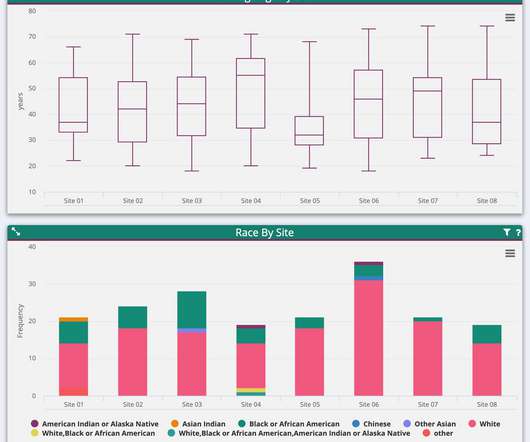
pharmaphorum
SEPTEMBER 11, 2020
Yet, for clinical trials worldwide, real-time access to data is not the norm – and that lack can create a cascading set of issues affecting every aspect of a study. What does this mean for clinical trials? Analysts at Pharma Intelligence decided to find out, surveying professionals involved in clinical data management.

pharmaphorum
JANUARY 10, 2025
A major population proteomics study using UK Biobank patient samples backed by 14 pharma companies has launched today.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content